Safety, pharmacokinetics, preliminary efficacy, pharmacodynamics, and immunogenicity of QX002N, an anti-IL-17A monoclonal antibody, after short-term treatment of active ankylosing spondylitis
- PMID: 40390042
- PMCID: PMC12090559
- DOI: 10.1186/s40360-025-00885-4
Safety, pharmacokinetics, preliminary efficacy, pharmacodynamics, and immunogenicity of QX002N, an anti-IL-17A monoclonal antibody, after short-term treatment of active ankylosing spondylitis
Abstract
Background: To investigate the safety, pharmacokinetics, preliminary efficacy, pharmacodynamics, and immunogenicity of QX002N, an interleukin-17 A monoclonal antibody, in Chinese patients with active ankylosing spondylitis (AS).
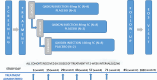
Methods: In this phase 1b, double-blind, placebo-controlled, multiple ascending dose study, eligible patients with active AS were randomized into three dose (40, 80, or 160 mg) cohorts, with a 4:1 ratio in each cohort to subcutaneously receive either QX002N or a placebo once every 2 weeks with six doses in total. All patients were followed for 14 weeks (98 days) after the last dose. The primary endpoints were the safety and pharmacokinetics of QX002N, and the secondary endpoints included its preliminary efficacy, pharmacodynamics, and immunogenicity.
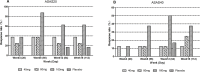
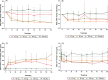
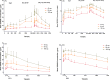
Results: Thirty patients (n = 10 in each cohort) were included, with 24 receiving QX002N and 6 receiving a placebo. A total of 85 adverse drug reactions, predominantly Grade 1-2, were identified in 20 out of 24 patients (83.3%) who took QX002N. The exposure to QX002N increased proportionally with the dose escalating from 40 mg to 160 mg. Patients taking 160 mg QX002N achieved higher response rates (ASAS20: 87.6% at Week 8 [Day 56]); ASAS40: 50.0% at Week 12 [Day 78]), than those taking 40-mg or 80-mg QX002N. An increase in interleukin-17 A and a decrease in interleukin-6 levels in the serum, with decreases in the erythrocyte sedimentation rate and high-sensitivity C-reactive protein levels, were observed. Anti-drug antibodies were detected in only one of 24 patients taking QX002N.
Conclusions: Subcutaneous administration of QX002N demonstrates a favorable safety profile, with linear pharmacokinetic characteristics. Promising clinical responses in pharmacodynamics and preliminary efficacy have been observed. Immunogenicity does not appear to be a concern.
Trial registration: This study was registered with Chinadrugtrials.org.cn (CTR20201277), the data of registration was in 20 Jul, 2020.
Keywords: Anti-IL-17A monoclonal antibody; Efficacy; Immunogenicity; Pharmacokinetics; QX002N; Safety.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethical approval: The study was approved by Ethics Committee of the First Hospital of Jilin University (approval number: 20Y116-001) and conducted in accordance with the Declaration of Helsinki and the Guidelines for Good Clinical Practice. Written informed consent was obtained from each participant before the study commenced. Consent to participate: All participants provided written informed consent for this study. Competing interests: The authors declare no competing interests.
Figures
References
-
- Ward MM, Deodhar A, Gensler LS, Dubreuil M, Yu D, Khan MA, Haroon N, Borenstein D, Wang R, Biehl A, et al. 2019 Update of the American college of rheumatology/spondylitis association of America/spondyloarthritis research and treatment network recommendations for the treatment of ankylosing spondylitis and nonradiographic axial spondyloarthritis. Arthritis Rheumatol. 2019;71:1599–613. 10.1002/art.41042. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

