No significant drug-drug interaction between oral TAF-based PrEP and feminizing hormone therapy among transgender women in Thailand: the iFACT-3 study
- PMID: 40390323
- PMCID: PMC12089648
- DOI: 10.1002/jia2.26502
No significant drug-drug interaction between oral TAF-based PrEP and feminizing hormone therapy among transgender women in Thailand: the iFACT-3 study
Abstract
Introduction: Concerns regarding potential drug-drug interaction (DDI) between feminizing hormone therapy (FHT) and HIV pre-exposure prophylaxis (PrEP) may hinder PrEP use among transgender women. We assessed the potential DDI between FHT and emtricitabine-tenofovir alafenamide (F/TAF)-based PrEP among transgender women.
Methods: Transgender women without HIV who never underwent orchiectomy were enrolled between January and February 2022. Oral FHT (oestradiol valerate 2 mg and cyproterone acetate 25 mg) was initiated at baseline and continued until week 9, while oral PrEP (F/TAF 200/25 mg) was initiated at week 3 and continued until week 12. Intensive blood sampling was performed at weeks 3 and 9 to assess the impact of PrEP on FHT; and weeks 9 and 12 to assess the impact of FHT on PrEP. Pharmacokinetics (PKs) of plasma oestradiol (E2), TAF, tenofovir (TFV) and emtricitabine (FTC); urine TFV and FTC; and tenofovir-diphosphate (TFV-DP) and emtricitabine-triphosphate (FTC-TP) in peripheral blood mononuclear cells (PBMCs) and rectal tissues were assessed.
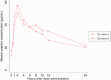
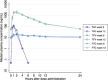
Results: Eighteen participants completed all PK visits. No significant differences in PK parameters for plasma E2, TAF and TFV were observed with FHT and F/TAF administration. The geometric mean of FTC AUC0-24 at week 9 was 9% lower than at week 12, but the 90% CI (0.88-0.95) remained within the 80-125% range. There were no significant differences in PBMCs and rectal tissues TFV-DP and FTC-TP concentrations when F/TAF was administered with FHT.
Conclusions: No bidirectional clinically significant DDI between FHT and F/TAF-based PrEP was observed across systemic and local tissue anatomical compartments, supporting the use of oral F/TAF-based PrEP among transgender women.
Clinical trial number: NCT04590417.
Keywords: HIV prevention; Thailand; drug−drug interactions; feminizing hormone therapy; pre‐exposure prophylaxis; transgender women.
© 2025 The Author(s). Journal of the International AIDS Society published by John Wiley & Sons Ltd on behalf of the International AIDS Society.
Conflict of interest statement
All authors declare no competing interests related to this work.
Figures

References
-
- Baral SD, Poteat T, Stromdahl S, Wirtz AL, Guadamuz TE, Beyrer C. Worldwide burden of HIV in transgender women: a systematic review and meta‐analysis. Lancet Infect Dis. 2013;13(3):214–22. - PubMed
-
- Coleman E, Bockting W, Botzer M, Cohen‐Kettenis P, DeCuypere G, Feldman J, et al. Standards of care (SOC) for the health of transsexual, transgender, and gender nonconforming people. Georgia: World Professional Association Transgender Health; 2011.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

