Vaginal Royal Jelly for Vulvovaginal Candidiasis Treatment: A Randomized Clinical Trial
- PMID: 40390812
- PMCID: PMC12087933
- DOI: 10.4103/abr.abr_69_24
Vaginal Royal Jelly for Vulvovaginal Candidiasis Treatment: A Randomized Clinical Trial
Abstract
Background: Vulvovaginal Candidiasis (VVC) is a prevalent and complex health issue in women. With the rising resistance to synthetic antifungal medications, there is a growing emphasis on exploring natural remedies that minimize disruption to the normal vaginal flora. The goal of this study is to evaluate the efficacy of vaginal Royal Jelly (RJ) compared with clotrimazole for VVC treatment.
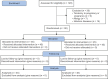
Materials and methods: This single-blind randomized clinical trial was conducted from 2020 to 2022 at Iran University of Medical Sciences hospitals, involving a total of 90 patients diagnosed with vaginal candidiasis. Positive vaginal culture individuals were randomly allocated to receive either vaginal 1000 mg RJ soft gel capsules or (2%) Clotrimazole cream (45 participants per group). Clinical signs, symptoms, and laboratory tests were statistically compared at baseline and ten days post-treatment. The data were analyzed using the independent t-tests, Mann-Whitney, Chi-square, and Wilcoxon tests through SPSS version 22.
Results: After 10 days of treatment, both groups demonstrated a significant decrease in Candida culture results (P < 0.001). RJ also positively impacted vaginal symptoms such as itching, discharge, and burning. Moreover, most patients in both groups became asymptomatic post-treatment.
Conclusions: The present study points out the therapeutic effects of vaginal RJ, a novel natural drug for improving VVC.
Keywords: Clotrimazole; royal jelly; vulvovaginal candidiasis.
Copyright: © 2025 Advanced Biomedical Research.
Conflict of interest statement
There are no conflicts of interest.
Figures
Similar articles
-
Comparison of the Effects of Nika Vaginal Cream with Clotrimazole Cream on Vaginal Candidiasis Symptoms: A Randomized Single-Blind Clinical Trial.Iran J Nurs Midwifery Res. 2021 Oct 22;26(6):521-525. doi: 10.4103/ijnmr.IJNMR_82_20. eCollection 2021 Nov-Dec. Iran J Nurs Midwifery Res. 2021. PMID: 34900651 Free PMC article.
-
Comparison of vaginal ointment of honey and clotrimazole for treatment of vulvovaginal candidiasis: A random clinical trial.J Mycol Med. 2017 Dec;27(4):494-500. doi: 10.1016/j.mycmed.2017.07.003. Epub 2017 Jul 29. J Mycol Med. 2017. PMID: 28760590 Clinical Trial.
-
The effect of curcumin-based and clotrimazole vaginal cream in the treatment of vulvovaginal candidiasis.J Family Med Prim Care. 2019 Dec 10;8(12):3920-3924. doi: 10.4103/jfmpc.jfmpc_584_19. eCollection 2019 Dec. J Family Med Prim Care. 2019. PMID: 31879636 Free PMC article.
-
Vulvovaginal candidiasis and vaginal microflora interaction: Microflora changes and probiotic therapy.Front Cell Infect Microbiol. 2023 Feb 3;13:1123026. doi: 10.3389/fcimb.2023.1123026. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 36816582 Free PMC article. Review.
-
Sertaconazole: a review of its use in the management of superficial mycoses in dermatology and gynaecology.Drugs. 2009;69(3):339-59. doi: 10.2165/00003495-200969030-00009. Drugs. 2009. PMID: 19275277 Review.
References
-
- Nyirjesy P, Brookhart C, Lazenby G, Schwebke J, Sobel JD. Vulvovaginal candidiasis: A review of the evidence for the 2021 centers for disease control and prevention of sexually transmitted infections treatment guidelines. Clin Infect Dis. 2022;74:S162–s8. - PubMed
-
- Sasani E, Rafat Z, Ashrafi K, Salimi Y, Zandi M, Soltani S, et al. Vulvovaginal candidiasis in Iran: A systematic review and meta-analysis on the epidemiology, clinical manifestations, demographic characteristics, risk factors, etiologic agents and laboratory diagnosis. Microb Pathog. 2021;154:104802. - PubMed
LinkOut - more resources
Full Text Sources