TNFα Receptor 1 and Not Receptor 2 Affect Annulus Fibrosus and Nucleus Pulposus Response to Cytokine Challenge in a Rat Model
- PMID: 40391206
- PMCID: PMC12086812
- DOI: 10.1002/jsp2.70070
TNFα Receptor 1 and Not Receptor 2 Affect Annulus Fibrosus and Nucleus Pulposus Response to Cytokine Challenge in a Rat Model
Abstract
Background: Painful intervertebral disc (IVD) degeneration (IVDD) involves chronic inflammation. Developing translational immunomodulatory strategies for IVDD is a priority with tumor necrosis factor alpha (TNFα) signaling an important target. TNFα binds to 2 receptors (TNFRs), with TNFR1 signaling promoting catabolism and apoptosis and TNFR2 signaling promoting anabolism and proliferation.
Methods: This study developed translational strategies to evaluate and modulate TNFR1 and TNFR2 signaling in rat in vivo and in vitro IVDD models. We used blocking antibodies, the TNFR2-activator Atsttrin, and small molecule inhibitors of TNFR1 to discern distinct TNFR1 and TNFR2-effects on annulus fibrosus (AF) and nucleus pulposus (NP) cells and to identify effective strategies for modulating specific TNFRs.
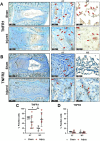
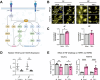
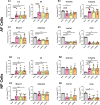
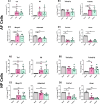
Results: TNFR1 was significantly increased with IVDD in vivo in the NP while TNFR2 was unaffected with very faint staining. TNFR1-specific small molecule inhibitors were effective in reducing catabolic effects of TNFα, highlighting the efficacy of this small molecule strategy for TNFR1 signaling modulation. Meanwhile, TNFR1 and TNFR2 inhibition in vitro was not effective with blocking antibodies on NP or AF cells, likely due to species-specificity of available blocking antibodies. Further, TNFR2 activation with Atsttrin was similarly ineffective, likely due to extremely low TNFR2 levels in both AF and NP cells.
Conclusions: TNFα receptor-specific signaling is important in rat IVDD in vivo and in vitro. TNFR1 inhibition was more effective with small molecules than using blocking antibodies. Low levels of TNFR2 in rat AF and NP cells and lack of efficacy of TNFR2-activator Atsttrin suggest native AF and NP cells have little capacity for TNFR2-dependent IVD repair.
Keywords: Atsttrin; Cell Culture; ErythrosineB; Physcion‐8‐O‐β‐D‐monoglucoside; inflammation; intervertebral disc degeneration; small molecules; tumor necrosis factor alpha.
© 2025 The Author(s). JOR Spine published by Wiley Periodicals LLC on behalf of Orthopaedic Research Society.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
TNFR1-mediated senescence and lack of TNFR2-signaling limit human intervertebral disc cell repair potential in degenerative conditions.Osteoarthritis Cartilage. 2025 Jul;33(7):874-887. doi: 10.1016/j.joca.2025.02.791. Epub 2025 Mar 24. Osteoarthritis Cartilage. 2025. PMID: 40139648
-
Therapeutic effects of PDGF-AB/BB against cellular senescence in human intervertebral disc.Elife. 2025 Jul 16;13:RP103073. doi: 10.7554/eLife.103073. Elife. 2025. PMID: 40668091 Free PMC article.
-
Distinctive roles of tumor necrosis factor receptor type 1 and type 2 in a mouse disc degeneration model.J Orthop Translat. 2021 Dec 3;31:62-72. doi: 10.1016/j.jot.2021.11.003. eCollection 2021 Nov. J Orthop Translat. 2021. PMID: 34934623 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

