Microwave-assisted synthesis of a zirconium-based MOF as an efficient catalyst for one-pot synthesis of xanthene derivatives: in silico study as a potential anti-HIV RNA
- PMID: 40391364
- PMCID: PMC12086817
- DOI: 10.1039/d5ra02959g
Microwave-assisted synthesis of a zirconium-based MOF as an efficient catalyst for one-pot synthesis of xanthene derivatives: in silico study as a potential anti-HIV RNA
Abstract
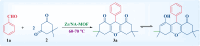
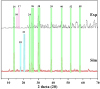
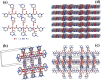
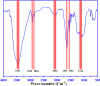
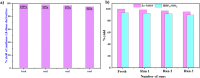
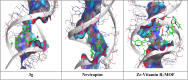
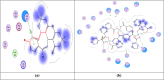
Firstly, construction and characterization of a novel Zr/VitB3 metal organic framework have been described. The physicochemical analysis and morphological properties of Zr-MOF has been accomplished by IR, SEM, EDX, TEM, and XRD. Cyclic diketones and various aromatic aldehydes were efficiently and ecologically benignly condensed in a single pot, yielding a range of tetrahydroxanthenediones in good to excellent yields. The utilization of a heterogeneous catalyst, solventless conditions, and a straightforward process that is atom-economical and yields low E-factor values are some of the benefits of this multicomponent reaction. The catalyst can be used up to three times and is completely recyclable. Advantages include a clean methodology, high yield, and straightforward catalyst preparation. Additionally, the study investigates the potential binding interactions of Zr-MOF with the HIV-RNA major groove, revealing its exceptional stability and strong binding affinity. The binding energy score of the Zr-MOF with HIV-RNA was found to be remarkably low at -12.32 kcal mol-1, indicating its potential to significantly outperform the reference molecule, nevirapine, which showed a higher E-score of -4.98 kcal mol-1. Xanthene derivatives were also evaluated for their binding affinity to the viral major groove, with energy scores ranging from -5.55 to -6.40 kcal mol-1, further indicating a promising potential for anti-HIV drug design. These findings underscore the potential of Zr-MOF and xanthene derivatives as potent candidates for HIV treatment, surpassing the reference molecule in terms of binding strength.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures




Similar articles
-
Synthesis and characterization of Co-MOF@Ag2O nanocomposite and its application as a nano-organic catalyst for one-pot synthesis of pyrazolopyranopyrimidines.Sci Rep. 2023 Oct 15;13(1):17500. doi: 10.1038/s41598-023-44667-6. Sci Rep. 2023. PMID: 37840041 Free PMC article.
-
Bi Metal-Organic Framework (Ce/Ni-BTC) as Heterogeneous Catalyst for the Green Synthesis of Substituted Chromeno[4, 3-b]quinolone under Solvent Free Condition.Curr Org Synth. 2021;18(5):475-482. doi: 10.2174/1570179418666210122100240. Curr Org Synth. 2021. PMID: 33480346
-
Application of novel metal-organic framework [Zr-UiO-66-PDC-SO3H]FeCl4 in the synthesis of dihydrobenzo[g]pyrimido[4,5-b]quinoline derivatives.RSC Adv. 2022 Mar 23;12(15):9058-9068. doi: 10.1039/d1ra08710j. eCollection 2022 Mar 21. RSC Adv. 2022. PMID: 35424891 Free PMC article.
-
Zr-Based Metal-Organic Frameworks for Green Biodiesel Synthesis: A Minireview.Bioengineering (Basel). 2022 Nov 17;9(11):700. doi: 10.3390/bioengineering9110700. Bioengineering (Basel). 2022. PMID: 36421101 Free PMC article. Review.
-
Indium-Organic Framework with soc Topology as a Versatile Catalyst for Highly Efficient One-Pot Strecker Synthesis of α-aminonitriles.ACS Appl Mater Interfaces. 2021 Nov 10;13(44):52023-52033. doi: 10.1021/acsami.1c09074. Epub 2021 Jul 1. ACS Appl Mater Interfaces. 2021. PMID: 34210117 Review.
References
-
- Chibale K. Visser M. van Schalkwyk D. Smith P. J. Saravanamuthu A. Fairlamb A. H. Tetrahedron. 2003;59:2289–2296.
-
- Maia M. Durães F. Resende D. I. Szemerédi N. Gales L. Martins-da-Costa P. Pinto M. Spengler G. Sousa E. Drugs and Drug Candidates. 2022;1:29–42.
-
- Zhao X. Wang Y. Gao X. Gao X. Wang M. Huang H. Liu B. Chin. Chem. Lett. 2025;36:109901.
LinkOut - more resources
Full Text Sources

