Sustained intraocular pressure-lowering effect and biocompatibility of a single subconjunctival administration of hydrogel-encapsulated nano-brinzolamide
- PMID: 40392392
- PMCID: PMC12092503
- DOI: 10.1007/s10856-025-06896-1
Sustained intraocular pressure-lowering effect and biocompatibility of a single subconjunctival administration of hydrogel-encapsulated nano-brinzolamide
Abstract
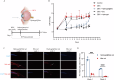
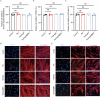
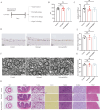
Brinzolamide is a widely used treatment for glaucoma, but its effectiveness relies on at least twice-daily dosing, which can be challenging for patient adherence. To overcome this limitation, we developed an injectable hydrogel-based delivery system designed to maintain therapeutic drug levels with a single administration. This approach aims to simplify treatment and improve clinical outcomes. Brinzolamide-loaded polyethylene glycol poly (lactic-co-glycolic acid) (PEG-PLGA) nanoparticles were encapsulated within a hydrogel synthesized through the crosslinking of oxidized hyaluronic acid (OHA) and carboxymethyl chitosan (CMC). In vitro studies were conducted to assess the nanoparticles' characterization, release profile, and biocompatibility. In a steroid-induced high intraocular pressure (IOP) mouse model, the efficacy of a single subconjunctival injection in lowering IOP was evaluated. Additionally, both cellular and animal biocompatibility were assessed. The brinzolamide-loaded hydrogel system (Hydrogel@Brz) contained nanoparticles with an average diameter of 40.76 nm, exhibiting a stable size distribution and a spherical morphology. The hydrogel demonstrated excellent injectability, self-healing properties, and a porous structure conducive to nanoparticle encapsulation. In vitro release studies revealed a sustained drug release of 86% over 14 days. No cytotoxicity was observed in human primary trabecular meshwork cells (HTMCs), human Tenon's capsule fibroblasts (HTFs), or the retinal ganglion cell line R28. In vivo, a single injection led to a prolonged IOP reduction lasting up to 21 days. No signs of drug toxicity were detected in ocular tissue sections, transverse optic nerve sections under transmission electron microscopy, or pathology slides of various organs. The brinzolamide-loaded hydrogel has demonstrated promising potential for sustained drug delivery and effective intraocular pressure reduction while maintaining good biocompatibility. However, further studies in larger animal models and long-term evaluations are needed to confirm its clinical applicability.
© 2025. The Author(s).
Conflict of interest statement
Compliance with ethical standards. Conflict of interest: The authors declare no competing interests.
Figures

Similar articles
-
Brinzolamide loaded core-shell nanoparticles for enhanced coronial penetration in the treatment of glaucoma.J Appl Biomater Funct Mater. 2020 Jan-Dec;18:2280800020942712. doi: 10.1177/2280800020942712. J Appl Biomater Funct Mater. 2020. PMID: 33151769
-
PLGA Nanoparticles as Subconjunctival Injection for Management of Glaucoma.AAPS PharmSciTech. 2017 Oct;18(7):2517-2528. doi: 10.1208/s12249-017-0710-8. Epub 2017 Feb 21. AAPS PharmSciTech. 2017. PMID: 28224390
-
Co-delivery of brinzolamide and miRNA-124 by biodegradable nanoparticles as a strategy for glaucoma therapy.Drug Deliv. 2020 Dec;27(1):410-421. doi: 10.1080/10717544.2020.1731861. Drug Deliv. 2020. PMID: 32133894 Free PMC article.
-
Preclinical overview of brinzolamide.Surv Ophthalmol. 2000 Jan;44 Suppl 2:S119-29. doi: 10.1016/s0039-6257(99)00108-3. Surv Ophthalmol. 2000. PMID: 10665514 Review.
-
Brinzolamide 1%/timolol 0.5%: safety and efficacy of a new fixed-combination IOP-lowering product for glaucoma.Curr Med Res Opin. 2010 Nov;26(11):2575-8. doi: 10.1185/03007995.2010.517718. Epub 2010 Sep 27. Curr Med Res Opin. 2010. PMID: 20868340 Review.
References
-
- Jonas JB, Aung T, Bourne RR, Bron AM, Ritch R, Panda-Jonas S. Glaucoma. Lancet. 2017;390:2183–93. - PubMed
-
- Holló G, Katsanos A, Boboridis K, Irkec M, Konstas A. G. P. Preservative-free prostaglandin analogs and prostaglandin/timolol fixed combinations in the treatment of glaucoma: efficacy, safety and potential advantages. Drugs. 2018;78:39–64. - PubMed
-
- Lusthaus JA, Goldberg I. Brimonidine and brinzolamide for treating glaucoma and ocular hypertension; a safety evaluation. Expert Opinion Drug Safety. 2017;16:1071–8. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

