Rhizosphere microorganisms mediate ion homeostasis in cucumber seedlings: a new strategy to improve plant salt tolerance
- PMID: 40394472
- PMCID: PMC12090690
- DOI: 10.1186/s12870-025-06699-0
Rhizosphere microorganisms mediate ion homeostasis in cucumber seedlings: a new strategy to improve plant salt tolerance
Abstract
Background: Soil salinization is a formidable challenge for vegetable production, primarily because of the detrimental effects of ion toxicity. Rhizosphere microorganisms promote plant growth and bolster salt tolerance, but the extent to which microbial communities can increase plant resilience by regulating ion homeostasis under salt stress remains underexplored. The goal of this study was to enrich microbial communities from the rhizosphere of salt-stressed cucumber seedlings and identify their impact on ion balance and plant growth under saline conditions.
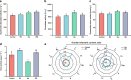
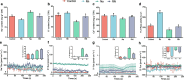
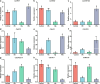
Results: Salt stress induced significant alterations in the composition, structure, and function of the root-associated microbial community. Compared with a 75 mM NaCl treatment alone, inoculation with salt-induced rhizosphere microorganisms (SiRMs) under the same conditions significantly increased the growth of cucumber seedlings; plant height increased by 61.3%, and the fresh weights of the shoots and roots increased by 45.3% and 38.9%, respectively. Moreover, superoxide dismutase (SOD) activity increased by 4.1%, and peroxidase (POD) activity and superoxide anion (O2·-) content decreased by 10.5% and 3.7%, respectively. In the roots, stems, and leaves of cucumber seedlings treated with SiRMs and 75 mM NaCl, the Na+ content was significantly reduced by 15.8%, 18.9%, and 9.7%, respectively. Conversely, the K+ content significantly increased by 32.7%, 16.9%, and 28.8%, respectively. Under salt stress conditions, inoculation with SiRMs significantly increased the rate of Na+ expulsion in the roots of cucumber seedlings by 18.3%, but the K+ expulsion rate decreased by 76.7%. These dynamic changes are attributed to the upregulation of genes such as CsHKT1, CsHAK5, and CsCHX18;4.
Conclusions: Enrichment with SiRMs played a pivotal role in maintaining ion homeostasis and significantly enhanced the salt tolerance of cucumber seedlings. These findings highlight the potential for microbial-assisted strategies to mitigate the adverse effects of soil salinity and provide valuable insights into the complex interplay between the microbial community and plant resilience from the perspective of ion balance.
Keywords: Cucumber; Ion homeostasis; Rhizosphere microorganisms; Salt stress.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures



Similar articles
-
Trichoderma harzianum mitigates salt stress in cucumber via multiple responses.Ecotoxicol Environ Saf. 2019 Apr 15;170:436-445. doi: 10.1016/j.ecoenv.2018.11.084. Epub 2018 Dec 13. Ecotoxicol Environ Saf. 2019. PMID: 30553921
-
Bacillus subtilis B55 degraded the ferulic acid and p-coumaric acid and changed the soil bacterial community in soils.J Appl Microbiol. 2024 Sep 2;135(9):lxae243. doi: 10.1093/jambio/lxae243. J Appl Microbiol. 2024. PMID: 39299920
-
Transcriptomic and functional characterization reveals CsHAK5;3 as a key player in K+ homeostasis in grafted cucumbers under saline conditions.Plant Sci. 2023 Jan;326:111509. doi: 10.1016/j.plantsci.2022.111509. Epub 2022 Oct 22. Plant Sci. 2023. PMID: 36283579
-
The CsGPA1-CsAQPs module is essential for salt tolerance of cucumber seedlings.Plant Cell Rep. 2020 Oct;39(10):1301-1316. doi: 10.1007/s00299-020-02565-5. Epub 2020 Jul 9. Plant Cell Rep. 2020. PMID: 32648011
-
Microbially Mediated Plant Salt Tolerance and Microbiome-based Solutions for Saline Agriculture.Biotechnol Adv. 2016 Nov 15;34(7):1245-1259. doi: 10.1016/j.biotechadv.2016.08.005. Epub 2016 Aug 30. Biotechnol Adv. 2016. PMID: 27587331 Review.
References
-
- Ji F, Wei SQ, Liu N, et al. Growth of cucumber seedlings in different varieties as affected by light environment. Int J Agric Biol Eng. 2020;13(5):73–8.
-
- Razzaq A, Ali A, Safdar LB, et al. Salt stress induces physiochemical alterations in rice grain composition and quality. J Food Sci. 2020;85(1):14–20. - PubMed
-
- Lu L, Huang M, Huang YX, et al. Mn3O4 nanozymes boost endogenous antioxidant metabolites in cucumber (Cucumis sativus) plant and enhance resistance to salinity stress. Environ Sci Nano. 2020;7(6):1692–703.
-
- Shahzad A, Ullah S, Dar AA, et al. Nexus on climate change: agriculture and possible solution to cope future climate change stresses. Environ Sci Pollut Res. 2021;28:14211–32. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

