Independent and synergistic roles of MEK-ERK1/2 and PKC pathways in regulating functional changes in vascular tissue following flow cessation
- PMID: 40395439
- PMCID: PMC12090243
- DOI: 10.1016/j.jmccpl.2025.100300
Independent and synergistic roles of MEK-ERK1/2 and PKC pathways in regulating functional changes in vascular tissue following flow cessation
Abstract
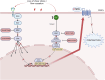
Background: The MEK-ERK1/2 and PKC pathways play critical roles in regulating functional changes in tissues, but their interplay remains poorly understood. The vasculature provides an ideal model to study these pathways, particularly under conditions of flow cessation, which is highly relevant to ischemia and other cardiovascular diseases. This study examined the independent roles, additive effects, and time-dependent dynamics of MEK and PKC pathway inhibition in functional receptor upregulation.
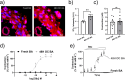
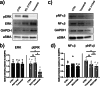
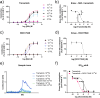
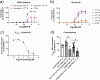
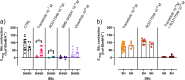
Methods: Rat basilar arteries were cultured for 48 h with selective inhibitors targeting MEK (Trametinib), PKC (RO-317549) and their downstream ERK (Ulixertinib) and NF-kB (BMS 345541). Functional changes in ETB receptor responses were assessed via wire myography following stimulation with Sarafotoxin 6c (S6c). Western blot analysis quantified ERK phosphorylation, and the effects of inhibitor timing and combination treatments were evaluated.
Results: MEK inhibition reduced ERK phosphorylation and ETB receptor-mediated contractility, whereas PKC inhibition had no effect on ERK phosphorylation but significantly reduced ETB receptor function. Combining MEK and PKC inhibitors produced an additive effect, resulting in greater suppression of functional changes compared to single treatments. At 6 h following flow cessation, PKC inhibition effectively suppressed ETB receptor function, while MEK inhibition had minimal effects when introduced at this delayed time point.
Conclusions: The MEK and PKC pathways independently drive functional changes in vascular tissue, particularly following flow cessation. MEK inhibition is effective early, while PKC inhibition remains effective when applied later. The additive effects observed with combined MEK and PKC inhibition indicate parallel and functionally independent pathway activation during ETB receptor upregulation.
Keywords: ETB upregulation; Flow cessation; Ischemia; MEK-ERK1/2 pathway; PKC pathway; Vasoconstriction.
© 2025 The Authors.
Conflict of interest statement
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Kristian Agmund Haanes reports financial support was provided by Lundbeck Foundation. Lars Edvinsson reports a relationship with Edvince AB, Sweden that includes: board membership and equity or stocks. Lars Edvinsson has patent #MEK INHIBITOR FOR TREATMENT OF STROKE - 20220273660 pending to Lars EDVINSSON. Lars Edvinsson has patent #Ischemic disorder or disease inhibitors -8273771 issued to Pronas Pharma AB. Lars Edvinsson has patent #ISCHEMIC DISORDER OR DISEASE INHIBITORS - 20100152248 pending to Pronas Pharma AB. All authors declare no conflicts of interest pertaining to the submitted work. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

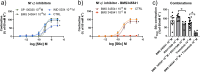
Similar articles
-
Increased endothelin-1-mediated vasoconstriction after organ culture in rat and pig ocular arteries can be suppressed with MEK/ERK1/2 inhibitors.Acta Ophthalmol. 2018 Aug;96(5):e619-e625. doi: 10.1111/aos.13651. Epub 2018 Jan 25. Acta Ophthalmol. 2018. PMID: 29369532
-
Myocardial ischemia-reperfusion enhances transcriptional expression of endothelin-1 and vasoconstrictor ETB receptors via the protein kinase MEK-ERK1/2 signaling pathway in rat.PLoS One. 2017 Mar 21;12(3):e0174119. doi: 10.1371/journal.pone.0174119. eCollection 2017. PLoS One. 2017. PMID: 28323857 Free PMC article.
-
Importance of ERK1/2 in upregulation of endothelin type B receptors in cerebral arteries.Br J Pharmacol. 2004 Aug;142(7):1155-61. doi: 10.1038/sj.bjp.0705803. Epub 2004 Jul 5. Br J Pharmacol. 2004. PMID: 15237095 Free PMC article.
-
Targeting ERK1/2 protein-serine/threonine kinases in human cancers.Pharmacol Res. 2019 Apr;142:151-168. doi: 10.1016/j.phrs.2019.01.039. Epub 2019 Feb 20. Pharmacol Res. 2019. PMID: 30794926 Review.
-
Endothelin in the pulmonary circulation with special reference to hypoxic pulmonary vasoconstriction.Scand Cardiovasc J Suppl. 1997;46:1-40. Scand Cardiovasc J Suppl. 1997. PMID: 9265559 Review.
References
-
- Edvinsson L. Cerebrovascular endothelin receptor upregulation in cerebral ischemia. Curr Vasc Pharmacol. 2009;7(1):26–33. - PubMed
-
- Macdonald R.L. Delayed neurological deterioration after subarachnoid haemorrhage. Nat Rev Neurol. 2014;10(1):44–58. - PubMed
-
- Liu, S., et al., Endothelin-1 Activates Endothelial Cell Nitric-oxide Synthase via Heterotrimeric G-protein βγ Subunit Signaling to Protein Kinase B/Akt *. J Biol Chem, 2003. 278(50): p. 49929–49935. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

