Gentiacaulein inhibits glucose transport to induce PRKAA1-mediated autophagy to clear amyloid beta and associated inflammation in primary astrocytes
- PMID: 40395536
- PMCID: PMC11864677
- DOI: 10.1080/27694127.2023.2296209
Gentiacaulein inhibits glucose transport to induce PRKAA1-mediated autophagy to clear amyloid beta and associated inflammation in primary astrocytes
Abstract
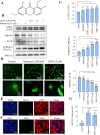
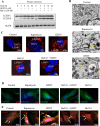
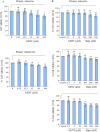
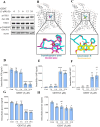
Being present in substantial numbers, astrocytes play an indispensable role in maintaining homeostasis in the brain. However, their positive or negative involvement in pathological conditions in the brain has not been explored much. In recent years, an emerging thought of targeting astrocytes for the resolution of neurodegenerative diseases has gained momentum. In this study, we have attempted to explore the likelihood of targeting astrocytes by using a natural compound, gentiacaulein (GENT), for clearance of amyloid-β (Aβ) through autophagy and amelioration of neuroinflammation associated with Aβ. We found that GENT treatment of astrocytes hampered the transport of glucose across the cell membrane, which resulted in a reduction in ATP production. With increased treatment time, AMP: ATP ratio was increased significantly, which caused the induction of PRKAA1-mediated autophagy. We further show that increased autophagy considerably enhanced the clearance of amyloid-β by astrocytes. GENT reduced the Aβ mediated inflammation by inhibiting the nuclear translocation of NF-κB and decreased the release of inflammatory cytokines TNF-α and IL-6. The role of PRKAA1 in GENT-induced autophagy and anti-inflammatory activity was confirmed when its knockdown reversed these effects. Our data suggest that targeting astrocytes can be a good strategy to prevent/treat Alzheimer's disease.
Keywords: Alzheimer disease; Amyloid-β; Astrocytes; Autophagy; Gentiacaulein; Neuroinflammation; PRKAA1/AMPK.
© 2023 The Author(s). Published by Informa UK Limited, trading as Taylor & Francis Group.
Conflict of interest statement
Authors declare no conflict of interest.
Figures



Similar articles
-
Fluvoxamine maleate alleviates amyloid-beta load and neuroinflammation in 5XFAD mice to ameliorate Alzheimer disease pathology.Front Immunol. 2024 Jul 29;15:1418422. doi: 10.3389/fimmu.2024.1418422. eCollection 2024. Front Immunol. 2024. PMID: 39136022 Free PMC article.
-
Clearance of β-amyloid mediated by autophagy is enhanced by MTORC1 inhibition but not AMPK activation in APP/PSEN1 astrocytes.Glia. 2024 Mar;72(3):588-606. doi: 10.1002/glia.24492. Epub 2023 Nov 27. Glia. 2024. PMID: 38009275
-
AMPK activation does not enhance autophagy in neurons in contrast to MTORC1 inhibition: different impact on β-amyloid clearance.Autophagy. 2021 Mar;17(3):656-671. doi: 10.1080/15548627.2020.1728095. Epub 2020 Feb 20. Autophagy. 2021. PMID: 32075509 Free PMC article.
-
Involvement of Astrocytes in Alzheimer's Disease from a Neuroinflammatory and Oxidative Stress Perspective.Front Mol Neurosci. 2017 Dec 19;10:427. doi: 10.3389/fnmol.2017.00427. eCollection 2017. Front Mol Neurosci. 2017. PMID: 29311817 Free PMC article. Review.
-
The role of astrocytes in amyloid production and Alzheimer's disease.Open Biol. 2017 Dec;7(12):170228. doi: 10.1098/rsob.170228. Open Biol. 2017. PMID: 29237809 Free PMC article. Review.
Cited by
-
Araloside A Induces Raf/MEK/ERK-Dependent Autophagy to Mitigate Alzheimer's and Parkinson's Pathology in Cellular and C. elegans Models.Mol Neurobiol. 2025 Jul 31. doi: 10.1007/s12035-025-05242-4. Online ahead of print. Mol Neurobiol. 2025. PMID: 40742404
References
-
- Herculano-Houzel S. The glia/neuron ratio: how it varies uniformly across brain structures and species and what that means for brain physiology and evolution. Glia 2014; 62:1377–91. - PubMed
LinkOut - more resources
Full Text Sources
