Riding a Vascular Time Train to Spatiotemporally Attenuate Thrombosis and Restenosis by Double Presentation of Therapeutic Gas and Biomacromolecules
- PMID: 40395762
- PMCID: PMC12087407
- DOI: 10.1002/EXP.70004
Riding a Vascular Time Train to Spatiotemporally Attenuate Thrombosis and Restenosis by Double Presentation of Therapeutic Gas and Biomacromolecules
Abstract
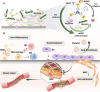
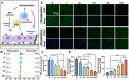
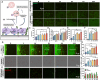
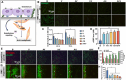
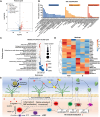
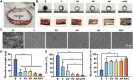
Endothelial injury is a common occurrence following stent implantation, often leading to complications such as restenosis and thrombosis. To address this issue, we have developed a multi-functional stent coating that combines a dopamine-copper (DA-Cu) base with therapeutic biomolecule modification, including nitric oxide (NO) precursor L-arginine, endothelial glycocalyx heparin, and endothelial cell (EC) catcher vascular endothelial growth factor (VEGF). In our stent coating, the incorporated Cu acts as a sustainable catalyst for converting endogenous NO donors into NO, and the immobilized arginine serves as a precursor for NO generation under the effect of endothelial nitric oxide synthase (eNOS). The presence of heparin endows the stent coating with anticoagulant ability and enhances eNOS activity, whilst rapid capture of EC by VEGF accelerates re-endothelialization. After in vivo implantation, the antioxidant elements and produced NO alleviate the inflammatory response, establishing a favorable healing environment. The conjugated VEGF contributes to the formation of a new and intact endothelium on the stent surface to counteract inappropriate vascular cell behaviors. The long-lasting NO flux inhibits smooth muscle cell (SMC) migration and prevents its excessive proliferation, reducing the risk of endothelial hyperplasia. This innovative coating enables the dual delivery of VEGF and NO to target procedural vascular repair phases: promoting rapid re-endothelialization, effectively preventing thrombosis, and suppressing inflammation and restenosis. Ultimately, this innovative coating has the potential to improve therapeutic outcomes following stent implantation.
Keywords: anti‐restenosis; anti‐thrombosis; biomacromolecule; re‐endothelialization; therapeutic gas.
© 2025 The Author(s). Exploration published by Henan University and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Gas station in blood vessels: An endothelium mimicking, self-sustainable nitric oxide fueling stent coating for prevention of thrombosis and restenosis.Biomaterials. 2023 Nov;302:122311. doi: 10.1016/j.biomaterials.2023.122311. Epub 2023 Aug 31. Biomaterials. 2023. PMID: 37677916
-
Immobilization of nano Cu-MOFs with polydopamine coating for adaptable gasotransmitter generation and copper ion delivery on cardiovascular stents.Biomaterials. 2019 Jun;204:36-45. doi: 10.1016/j.biomaterials.2019.03.007. Epub 2019 Mar 9. Biomaterials. 2019. PMID: 30875517
-
Vascular endothelial growth factor (VEGF)-eluting stents: in vivo effects on thrombosis, endothelialization and intimal hyperplasia.J Invasive Cardiol. 2003 Dec;15(12):688-92. J Invasive Cardiol. 2003. PMID: 14660819
-
Drug-eluting stents.Arch Cardiol Mex. 2006 Jul-Sep;76(3):297-319. Arch Cardiol Mex. 2006. PMID: 17091802 Review.
-
Vascular inflammation and repair: implications for re-endothelialization, restenosis, and stent thrombosis.JACC Cardiovasc Interv. 2011 Oct;4(10):1057-66. doi: 10.1016/j.jcin.2011.05.025. JACC Cardiovasc Interv. 2011. PMID: 22017929 Free PMC article. Review.
References
-
- (a) Palmerini T., Benedetto U., Biondi‐Zoccai G., et al., “Long‐Term Safety of Drug‐Eluting and Bare‐Metal Stents,” Journal of the American College of Cardiology 65 (2015): 2496–2507. - PubMed
- (b) Frieden T. R. and Jaffe M. G., “Saving 100 Million Lives by Improving Global Treatment of Hypertension and Reducing Cardiovascular Disease Risk Factors,” The Journal of Clinical Hypertension 20 (2018): 208–211. - PMC - PubMed
-
- (a) Yang Y., Gao P., Wang J., et al., “Endothelium‐Mimicking Multifunctional Coating Modified Cardiovascular Stents via a Stepwise Metal‐Catechol‐(Amine) Surface Engineering Strategy,” Research 2020 (2020): 9203906. - PMC - PubMed
- (b) Yang Z., Zhao X., Hao R., et al., “Bioclickable and Mussel Adhesive Peptide Mimics for Engineering Vascular Stent Surfaces,” PNAS 117 (2020): 16127–16137. - PMC - PubMed
-
- (a) Habib A. and Finn A. V., “Endothelialization of Drug Eluting Stents and Its Impact on Dual Anti‐platelet Therapy Duration,” Pharmacological Research 93 (2015): 22–27. - PMC - PubMed
- (b) Lee H., Dellatore S. M., Miller W. M., and Messersmith P. B., “Mussel‐Inspired Surface Chemistry for Multifunctional Coatings,” Science 318 (2007): 426–430. - PMC - PubMed
-
- (a) Straub A. C., Lohman A. W., Billaud M., et al., “Endothelial Cell Expression of Haemoglobin α Regulates Nitric Oxide Signalling,” Nature 491 (2012): 473–477. - PMC - PubMed
- (b) Li P., Cai W., Wang K., et al., “Selenium‐Functionalized Polycarbonate‐Polyurethane for Sustained In Situ Generation of Therapeutic Gas for Blood‐Contacting Materials,” Smart Materials in Medicine 3 (2022): 361–373.
LinkOut - more resources
Full Text Sources
