Causonis trifolia- based green synthesis of multifunctional silver nanoparticles for dual sensing of mercury and ferric ions, photocatalysis, and biomedical applications
- PMID: 40395792
- PMCID: PMC12091325
- DOI: 10.1039/d5ra01882j
Causonis trifolia- based green synthesis of multifunctional silver nanoparticles for dual sensing of mercury and ferric ions, photocatalysis, and biomedical applications
Abstract
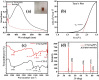
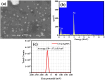
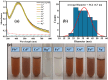
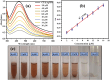
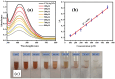
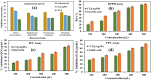
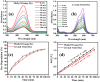
Health and environmental concerns are often raised by the development of antibiotic resistance and water contamination from various aquatic contaminants, including antibiotic residues, dyes, and heavy metal ions. This paper outlines a facile, affordable, and eco-friendly way to address these issues by green synthesis of silver nanoparticles (CT@AgNPs) under sunlight irradiation using Causonis trifolia leaf extract (CTLE), known for its medicinal properties. The greenly synthesized CT@AgNPs exhibited antioxidant, antibacterial, and photocatalytic properties and were an effective nanoprobe for the selective detection of Fe3+ and Hg2+ in water. CT@AgNPs were thoroughly examined using several sophisticated analytical methods, including FTIR, UV-vis spectroscopy, Scanning electron microscopy (SEM), Powder X-ray diffraction (PXRD), Energy dispersive X-ray (EDX), and Zeta potential (ZP). FTIR demonstrated the effective functionalization of CT@AgNPs with the polar leaf extract of Causonis trifolia. The optical properties of CT@AgNPs in solution were monitored using UV-vis spectrophotometric analysis. The synthesis of spherical shaped CT@AgNPs with a face-centered cubic geometry and a 12.7 nm average crystallite size was assessed by SEM and XRD, respectively. CT@AgNPs showed a potent antibacterial activity against Gram-positive bacteria (L. monocytogenes and S. epidermidis) and Gram-negative bacterial strains (P. aeruginosa and B. bronchiseptica). CT@AgNPs showed high sensitivity for colorimetric detection of Hg2+ and Fe3+ with a limit of detection of 1.04 μM and 47.57 μM, respectively in spiked water samples, highlighting their potential for use in environmental monitoring applications. CT@AgNPs showed remarkable antioxidant ability, assessed by DPPH, TFC, and TPC assays. On exposure to sunlight, CT@AgNPs also showed good photocatalytic capability by degradation of methyl orange (79%) and crystal violet (77%) with rate constant values of 0.0157 min-1, and 0.0150 min-1, respectively. This work demonstrates the potential of green route-synthesized AgNPs as efficient and sustainable materials for biomedical and environmental applications.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors have no known financial or non-financial interests to disclose.
Figures
References
-
- Sarma P. P. Barman K. Baruah P. K. Inorg. Chem. Commun. 2023;152:110676.
-
- Mishra N. Dhwaj A. Verma D. Prabhakar A. Anal. Chim. Acta. 2022;1205:339734. - PubMed
-
- Tamilselvan S. Soniya R. Vasantharaja R. Kannan M. Supriya S. Batvari B. P. D. Ramesh T. Govindaraju K. Environ. Res. 2022;212:113585. - PubMed
-
- Singh R. Mehra R. Walia A. Gupta S. Chawla P. Kumar H. Thakur A. Kaushik R. Kumar N. Int. J. Environ. Anal. Chem. 2023;103:1361–1376.
LinkOut - more resources
Full Text Sources
Miscellaneous

