Comparison between upper body and full underbody forced-air warming blanket in pediatric patients undergoing cardiovascular interventions under general anesthesia: a randomized controlled trial
- PMID: 40399788
- PMCID: PMC12093743
- DOI: 10.1186/s12871-025-03100-3
Comparison between upper body and full underbody forced-air warming blanket in pediatric patients undergoing cardiovascular interventions under general anesthesia: a randomized controlled trial
Abstract
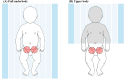
Background: Pediatric patients undergoing cardiovascular interventions outside the operating room are at high risk of perioperative hypothermia. We aimed to compare the effects of upper body and full underbody forced-air warming blankets on the time-weighted deviation of esophageal temperature outside the target range (36.5-37.5 °C) during general anesthesia.
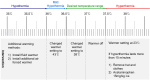
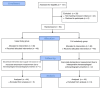
Methods: In this randomized controlled study, 88 children (age < 15 years) scheduled for elective cardiovascular interventions under general anesthesia were randomly assigned to the upper body (n = 44) or full underbody (n = 44) group. After the induction of anesthesia, warming blankets were applied and heated using a forced-air warmer to maintain an esophageal temperature of 36.5-37.5 °C. The primary outcome was the time-weighted average deviation of esophageal temperature outside the desired range, defined as the total deviation in temperature divided by the duration spent outside the target range. Secondary outcomes included use of additional warming or cooling methods, temperature trends, thermal comfort, and adverse events. Statistical comparisons were performed using t-tests or chi-square tests, with p < 0.05 considered significant.
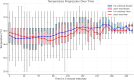
Results: The time-weighted averages of periods out of the desired temperature range were comparable between the two groups (upper body vs. full underbody, 0.213 ± 0.212 °C vs. 0.265 ± 0.277 °C; mean difference, 0.053; 95% confidence interval [CI], - 0.052 to 0.157; p = 0.318). The incidence of hyperthermia (> 37.5 °C) was 9.09% (upper body) and 0% (full underbody, p = 0.125). The duration of hypothermia (< 36.5 °C) was 58.82 ± 48.83 min (upper body) and 70.03 ± 53.20 min (full underbody; mean difference, 11.20 min; 95% CI, - 10.44 to 32.85; p = 0.318). The incidence rates of adverse events were 4.55% (upper body) and 15.91% (full underbody, p = 0.159).
Conclusions: Both warming methods showed comparable time-weighted averages of temperatures outside the desired range, suggesting similar effectiveness. However, careful monitoring is essential to mitigate the risks of hyperthermia and skin-related complications and ensure patient safety during pediatric cardiovascular interventions.
Trial registration number: NCT05349734 (registered at clinicaltrials, principal investigator: Hee-Soo Kim, registration date: April 26,2022).
Keywords: Cardiac intervention; Forced-air warming; Hypothermia; Pediatric anesthesia.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This prospective randomized controlled study was conducted at Seoul National University Children’s Hospital between May 2022 and January 2024. This study was approved by the Institutional Review Board of Seoul National University Hospital (approval no. H-2005-016-1121, approval date: April 19, 2022, chairperson: Hyun-Hoon Jung) and registered at http://clinicaltrials.gov (registration number: NCT05349734, principal investigator: Hee-Soo Kim, registration date: April 26, 2022) prior to patient enrolment. Written informed consent was obtained from the parents or legal guardians of all participants prior to study enrollment. Consent for publication: Not applicable. Presentation: None. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Underbody carbon-fiber contact warming versus underbody forced-air warming to prevent hypothermia during laparoscopic gynecologic surgery: A randomized trial.Rev Esp Anestesiol Reanim (Engl Ed). 2025 May;72(5):501689. doi: 10.1016/j.redare.2025.501689. Epub 2025 Feb 15. Rev Esp Anestesiol Reanim (Engl Ed). 2025. PMID: 39961536 Clinical Trial.
-
Comparative effects of warming systems applied to different parts of the body on hypothermia in adults undergoing abdominal surgery: A systematic review and network meta-analysis of randomized controlled trials.J Clin Anesth. 2023 Oct;89:111190. doi: 10.1016/j.jclinane.2023.111190. Epub 2023 Jun 28. J Clin Anesth. 2023. PMID: 37390588
-
Interventions to prevent hypothermia at birth in preterm and/or low birth weight infants.Cochrane Database Syst Rev. 2018 Feb 12;2(2):CD004210. doi: 10.1002/14651858.CD004210.pub5. Cochrane Database Syst Rev. 2018. PMID: 29431872 Free PMC article.
-
Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis.Cochrane Database Syst Rev. 2020 Oct 19;10(10):CD012859. doi: 10.1002/14651858.CD012859.pub2. Cochrane Database Syst Rev. 2020. PMID: 33075160 Free PMC article.
-
Interventions for infantile haemangiomas of the skin.Cochrane Database Syst Rev. 2018 Apr 18;4(4):CD006545. doi: 10.1002/14651858.CD006545.pub3. Cochrane Database Syst Rev. 2018. PMID: 29667726 Free PMC article.
References
-
- Conway A, Kennedy W, Sutherland J. Inadvertent hypothermia after procedural sedation and analgesia in a cardiac catheterization laboratory: A prospective observational study. J Cardiothorac Vasc Anesth. 2015;29(5):1285–90. - PubMed
-
- Bajwa SJS, Swati M. Perioperative hypothermia in pediatric patients: diagnosis, prevention and management. Anaesth Pain Intensive Care 2019:97–100.
-
- Pearce B, Christensen R, Voepel-Lewis T. Perioperative hypothermia in the pediatric population: prevalence, risk factors and outcomes. J Anesthe Clin Res. 2010;1(1):1–4.
-
- Roe CF, Santulli TV, Blair CS. Heat loss in infants during general anesthesia and operations. J Pediatr Surg. 1966;1(3):266–74. - PubMed
-
- Wang J-f. Deng X-m: inadvertent hypothermia: a prevalent perioperative issue that remains to be improved. Anesthesiology Perioperative Sci. 2023;1(3):24.
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

