Ectonucleotidases and purinergic receptors in mouse prostate gland
- PMID: 40400995
- PMCID: PMC12089222
- DOI: 10.62347/NGQZ2940
Ectonucleotidases and purinergic receptors in mouse prostate gland
Abstract
Objectives: Extracellular ATP/ADP and its metabolite adenosine play crucial roles in cellular signaling by interacting with P2 and P1/adenosine receptors. These signaling molecules are regulated by ectonucleotidases, which convert ATP/ADP into adenosine. While recent studies suggest impaired ATP hydrolysis in the aging prostate, the expression and function of ectonucleotidases and purinergic receptors in the prostate gland remain unclear. This study aims to characterize the expression patterns of purinergic enzymes and receptors in the mouse prostate and investigate their functional implications.
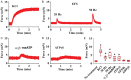
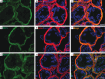
Methods: Mouse prostate glands were isolated and analyzed using immunofluorescent staining and microscopy imaging with specific antibodies to detect purinergic enzymes and receptors. Functional studies were conducted to assess prostate smooth muscle contraction in response to purinergic agonists, particularly α,β-meATP and ATPγS.
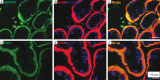
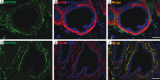
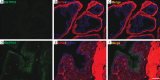
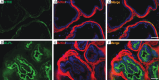
Results: Our analysis revealed distinct expression patterns of purinergic enzymes and receptors in the prostate: Ectonucleoside triphosphate diphosphohydrolase 1 (ENTPD1) and P2X1 receptors were predominantly localized in prostate smooth muscle cells, ENTPD2 and ecto-5'-nucleotidase (NT5E) in prostate interstitial cells, and alkaline phosphatase (ALPL) in prostate epithelial cells. Notably, ENTPD1 was identified as a key ectonucleotidase expressed in mouse prostate smooth muscle cells. Functionally, P2X1-mediated smooth muscle contraction was triggered by α,β-meATP. However, ATPγS induced contraction even after P2X1 desensitization, suggesting the involvement of additional P2Y receptors. Further analysis confirmed the presence of P2Y1, P2Y2, and P2Y11 receptors in mouse prostate smooth muscle, likely mediating the ATPγS-induced contraction.
Conclusions: This study provides a comprehensive characterization of purinergic signaling components in the mouse prostate. The identification of ENTPD1 in smooth muscle cells and the functional role of multiple P2Y receptors in smooth muscle contraction highlight potential regulatory mechanisms of prostate function. These findings lay the groundwork for future research on purinergic signaling in prostate physiology and its potential implications in age-related dysfunction, both in rodents and humans.
Keywords: P2Y receptor; Purinergic signaling; alkaline phosphatase; mouse prostate; smooth muscle contractility.
AJCEU Copyright © 2025.
Conflict of interest statement
None.
Figures
Similar articles
-
P2 receptors in the murine gastrointestinal tract.Neuropharmacology. 2002 Dec;43(8):1313-23. doi: 10.1016/s0028-3908(02)00294-0. Neuropharmacology. 2002. PMID: 12527481
-
Unusual absence of endothelium-dependent or -independent vasodilatation to purines or pyrimidines in the rat renal artery.Kidney Int. 2003 Oct;64(4):1389-97. doi: 10.1046/j.1523-1755.2003.00233.x. Kidney Int. 2003. PMID: 12969158
-
The ecto-nucleotidase NTPDase1 differentially regulates P2Y1 and P2Y2 receptor-dependent vasorelaxation.Br J Pharmacol. 2010 Feb 1;159(3):576-85. doi: 10.1111/j.1476-5381.2009.00566.x. Epub 2010 Jan 8. Br J Pharmacol. 2010. PMID: 20067476 Free PMC article.
-
Purinergic transmission in blood vessels.Auton Neurosci. 2015 Sep;191:48-66. doi: 10.1016/j.autneu.2015.04.007. Epub 2015 Apr 25. Auton Neurosci. 2015. PMID: 26004513 Review.
-
Purinergic regulation of pulmonary vascular tone.Purinergic Signal. 2024 Dec;20(6):595-606. doi: 10.1007/s11302-024-10010-5. Epub 2024 May 7. Purinergic Signal. 2024. PMID: 38713328 Free PMC article. Review.
References
-
- Chughtai B, Forde JC, Thomas DD, Laor L, Hossack T, Woo HH, Te AE, Kaplan SA. Benign prostatic hyperplasia. Nat Rev Dis Primers. 2016;2:16031. - PubMed
-
- Ng M, Leslie SW, Baradhi KM. Benign prostatic hyperplasia. In: editors. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: stephen Leslie declares no relevant financial relationships with ineligible companies. Disclosure: Krishna Baradhi declares no relevant financial relationships with ineligible companies. 2024.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
