Dysregulated Tricarboxylic Acid Cycle Metabolism Is Associated With Right Ventricular Maladaptation in Pulmonary Vascular Disease
- PMID: 40401597
- PMCID: PMC12229207
- DOI: 10.1161/JAHA.124.041127
Dysregulated Tricarboxylic Acid Cycle Metabolism Is Associated With Right Ventricular Maladaptation in Pulmonary Vascular Disease
Abstract
Background: Right ventricular (RV) maladaptation to elevated pulmonary afterload is the primary determinant of outcomes in pulmonary artery (PA) hypertension; however, the pathobiological mechanisms underlying RV decompensation remain poorly understood.
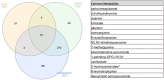
Methods: We performed global untargeted metabolomics on plasma from 55 patients who underwent gold-standard RV-PA coupling measurements using multibeat pressure volume loop assessment in a single-center cohort and from 1027 patients with coupling surrogate measurements in a larger multicenter cohort, the PVDOMICS (Pulmonary Vascular Disease Phenomics) study. Age and sex-adjusted linear regression was performed to identify associations between metabolites and coupling metrics. Additionally, we performed a metabolic flux analysis using gene expression data from RV tissue in an independent cohort of 32 patients. Partial least squares-discriminant analysis was used to identify metabolites and reactions characteristic of the decompensated RV.
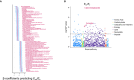
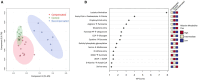
Results: RV-PA coupling was negatively associated with tricarboxylic acid (TCA) cycle intermediate levels. Specifically, plasma α-ketoglutarate and fumarate were significantly associated with all coupling metrics in both cohorts. Metabolic flux analysis indicated that decompensated RVs exhibited aberrant TCA cycle activity, including reduced acetyl coenzyme A entry and increased lactate elimination, suggesting a shift from the TCA cycle toward glycolysis at the RV tissue level.
Conclusions: We identify an association between circulating TCA cycle intermediate levels and RV-PA uncoupling in 2 independent cohorts, and dysregulated TCA cycle metabolism in decompensated PA hypertension RVs, suggesting that aberrant TCA cycle metabolism could represent a hallmark of RV maladaptation in PA hypertension. Further study of this pathway is warranted to develop novel biomarkers of RV function or RV-targeted therapies.
Keywords: global untargeted metabolomics; right ventricular maladaptation; right ventricular–pulmonary artery coupling; tricarboxylic acid cycle.
Conflict of interest statement
Dr Tedford is Deputy Editor for the
Figures



References
-
- Vonk Noordegraaf A, Chin KM, Haddad F, Hassoun PM, Hemnes AR, Hopkins SR, Kawut SM, Langleben D, Lumens J, Naeije R. Pathophysiology of the right ventricle and of the pulmonary circulation in pulmonary hypertension: an update. Eur Respir J. 2019;53. doi: 10.1183/13993003.01900-2018 - DOI - PMC - PubMed
-
- Brener MI, Masoumi A, Ng VG, Tello K, Bastos MB, Cornwell WK, Hsu S, Tedford RJ, Lurz P, Rommel K‐P, et al. Invasive right ventricular pressure‐volume analysis: basic principles, clinical applications, and practical recommendations. Circ Heart Fail. 2022;15. doi: 10.1161/circheartfailure.121.009101 - DOI - PMC - PubMed
-
- Tedford RJ, Mudd JO, Girgis RE, Mathai SC, Zaiman AL, Housten‐Harris T, Boyce D, Kelemen BW, Bacher AC, Shah AA, et al. Right ventricular dysfunction in systemic sclerosis‐associated pulmonary arterial hypertension. Circ Heart Fail. 2013;6:953–963. doi: 10.1161/circheartfailure.112.000008 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

