Exosomal PD-L1 derived from hypoxia nasopharyngeal carcinoma cell exacerbates CD8+ T cell suppression by promoting PD-L1 upregulation in macrophages
- PMID: 40411570
- PMCID: PMC12103439
- DOI: 10.1007/s00262-025-04047-7
Exosomal PD-L1 derived from hypoxia nasopharyngeal carcinoma cell exacerbates CD8+ T cell suppression by promoting PD-L1 upregulation in macrophages
Abstract
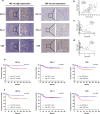
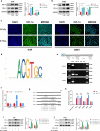
Immunotherapy targeting the programmed death ligand-1/programmed cell death protein-1 (PD-L1/PD-1) pathway exhibits limited effectiveness in individuals with recurrent and metastatic nasopharyngeal carcinoma (NPC). Recent studies have noted that hypoxia within the tumor microenvironment (TME) triggers intricate interplay, termed "hypoxia-induced exosome-mediated communication", between cancer cells and various immune cells. However, the role of hypoxia in modulating the immunosuppressive environment and its implications on the efficacy of immunotherapy in NPC remains poorly understood. In this study, we found hypoxia inducible factor-1 (HIF-1α) was positively associated with increased PD-L1 levels and decreased CD8+ T cell infiltration, and correlated with a poor prognosis. Mechanistically, we demonstrated that hypoxia regulated the expression of PD-L1 in NPC cells and their exosomes by activating the binding of HIF-1α to the PD-L1 promoter. Meanwhile, using in vitro approaches, we found that macrophages could upregulate their PD-L1 expression through the phagocytosis of exosomal PD-L1 derived from NPC cells. Furthermore, we confirmed that PD-L1+ macrophages could induce CD8+ T cell exhaustion and reduce their proliferation. In conclusion, our study revealed that hypoxia (via HIF-1α) upregulated the expression of PD-L1 in exosomes derived from NPC cells, while macrophages induce the suppression of CD8+ T cells by phagocytosis of exosomal PD-L1. Targeting the PD-L1+ macrophages could potentially serve as a promising approach to augment the effectiveness of immune checkpoint blockade in NPC.
Keywords: Hypoxia; Immune escape; Macrophage; Nasopharyngeal carcinoma; PD-L1.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: The authors declare that there are no conflicts of interest regarding the publication of this paper. Ethical approval: This study was approved by the Ethics Committee of Nanfang Hospital of Southern Medical University (Approval no.: NFEC-2017-165). All participants provided written informed consent for the use of their samples and data in research. The study adhered to the Declaration of Helsinki and ensured participant confidentiality.
Figures





Similar articles
-
Targeting the LMP1-ALIX axis in EBV+ nasopharyngeal carcinoma inhibits immunosuppressive small extracellular vesicle secretion and boosts anti-tumor immunity.Cancer Commun (Lond). 2024 Dec;44(12):1391-1413. doi: 10.1002/cac2.12619. Epub 2024 Oct 14. Cancer Commun (Lond). 2024. PMID: 39402748 Free PMC article.
-
Nasopharyngeal cancer cell-derived exosomal PD-L1 inhibits CD8+ T-cell activity and promotes immune escape.Cancer Sci. 2022 Sep;113(9):3044-3054. doi: 10.1111/cas.15433. Epub 2022 Jul 12. Cancer Sci. 2022. PMID: 35598173 Free PMC article.
-
Hypoxic tumour-derived exosomal miR-1290 exacerbates the suppression of CD8+ T cells by promoting M2 macrophage polarization.Immunology. 2024 Dec;173(4):672-688. doi: 10.1111/imm.13853. Epub 2024 Aug 26. Immunology. 2024. PMID: 39183579
-
Cancer stem cell-derived exosomes in CD8+ T cell exhaustion.Int Immunopharmacol. 2024 Aug 20;137:112509. doi: 10.1016/j.intimp.2024.112509. Epub 2024 Jun 17. Int Immunopharmacol. 2024. PMID: 38889509 Review.
-
Exosomal PD-L1: Roles in Tumor Progression and Immunotherapy.Trends Cancer. 2020 Jul;6(7):550-558. doi: 10.1016/j.trecan.2020.03.002. Epub 2020 Mar 29. Trends Cancer. 2020. PMID: 32610067 Free PMC article. Review.
Cited by
-
Targeting Exosomal PD-L1 as a New Frontier in Cancer Immunotherapy.Curr Issues Mol Biol. 2025 Jul 8;47(7):525. doi: 10.3390/cimb47070525. Curr Issues Mol Biol. 2025. PMID: 40728994 Free PMC article. Review.
References
-
- Wong K, Hui EP, Lo KW et al (2021) Nasopharyngeal carcinoma: an evolving paradigm. Nat Rev Clin Oncol 18(11):679–695 - PubMed
-
- Chen YP, Chan A, Le QT et al (2019) Nasopharyngeal carcinoma. Lancet 394(10192):64–80 - PubMed
-
- Fang W, Yang Y, Ma Y et al (2018) Camrelizumab (SHR-1210) alone or in combination with gemcitabine plus cisplatin for nasopharyngeal carcinoma: results from two single-arm, phase 1 trials. Lancet Oncol 19(10):1338–1350 - PubMed
MeSH terms
Substances
Grants and funding
- 20231800940662/Dongguan Science and Technology of Social Development Program
- 20231800940662/Dongguan Science and Technology of Social Development Program
- 20231800940662/Dongguan Science and Technology of Social Development Program
- 2021B1515140070/Guangdong Basic and Applied Basic Research Foundation
- 2021B1515140070/Guangdong Basic and Applied Basic Research Foundation
LinkOut - more resources
Full Text Sources
Research Materials

