Annexin A6 membrane repair protein protects against amyloid-induced dystrophic neurites and tau phosphorylation in Alzheimer's disease model mice
- PMID: 40411591
- PMCID: PMC12103342
- DOI: 10.1007/s00401-025-02888-1
Annexin A6 membrane repair protein protects against amyloid-induced dystrophic neurites and tau phosphorylation in Alzheimer's disease model mice
Abstract
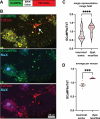
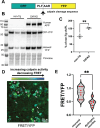
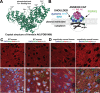
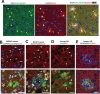
In Alzheimer's disease, accumulation of amyloid-β (Aβ) peptide is thought to cause formation of neurofibrillary tangles composed of hyperphosphorylated tau protein, which correlates with neuronal loss and cognitive impairment, but the mechanism linking Aβ and tau pathologies is unknown. Dystrophic neurites, which surround Aβ plaques and accumulate phosphorylated tau and other proteins, may play a role in seeding and spreading of pathologic tau. Here, we investigate the novel hypothesis that improved membrane repair capacity decreases dystrophic neurite formation by protecting axons from Aβ-induced membrane damage. Using a ratiometric calcium sensor and a FRET-based calpain cleavage sensor, we demonstrate that dystrophic neurites in 5XFAD mice have elevated resting calcium levels and calpain activity because of putative membrane damage. Annexin A6, a plasma membrane repair in muscle and neurons, is present at plasma membrane of neurons and dystrophic neurites in murine and human brains. Overexpression of annexin A6 in brains of 5XFAD mice decreased size and quantity of dystrophic neurites and accumulation of phospho-tau181, an early biomarker of amyloid pathology. Phospho-tau231, another early amyloid biomarker, and phosphorylated of tau kinases, c-jun N-terminal kinase (JNK) and Calmodulin Kinase II (CaMKII) accumulate in dystrophic neurites in the brains of amyloid pathology mice and humans with AD, suggesting that dystrophic neurites are sites of active tau phosphorylation. Overexpression of dominant-negative annexin A6 in 5XFAD mice increased dystrophic neurites and phospho-tau181. Intracerebral injection of recombinant annexin A6 in 5XFAD and APP-NLGF knock-in mice resulted in localization of recombinant A6 to membranes of dystrophic neurites, suggesting therapeutic potential of recombinant annexin A6 for AD. In conclusion, dystrophic neurites have Aβ-induced membrane damage characterized by calcium elevation, calpain activation, and accumulation of tau kinases and phosphorylated tau. Overexpression of annexin A6 reduces dystrophic neurites and phospho-tau accumulation, suggesting that annexin A6-mediated membrane repair may represent a novel therapeutic approach for AD.
Keywords: Alzheimer’s disease; Amyloid pathology; Calcium dysregulation; Dystrophic neurites; Membrane repair; Tau phosphorylation.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: Northwestern University filed a US provisional patent application on behalf of KRS, ARD, EMM, and RV related to the use of annexins in treating neuronal cell membrane injury in September 2022.
Figures



References
-
- (2023) The Alzheimer’s Association, Alzheimer’s Disease: Facts and Figures https://www.alz.org/alzheimers-dementia/facts-figures - PubMed
-
- Alexander GC, Emerson S, Kesselheim AS (2021) Evaluation of aducanumab for alzheimer disease: scientific evidence and regulatory review involving efficacy, safety, and futility. JAMA 325:1717–1718. 10.1001/jama.2021.3854 - PubMed
-
- Alzheimer’s Association Calcium Hypothesis W (2017) Calcium hypothesis of Alzheimer’s disease and brain aging: a framework for integrating new evidence into a comprehensive theory of pathogenesis. Alzheimers Dement 13(178–182):e117. 10.1016/j.jalz.2016.12.006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

