Ultrasound guided transabdominal botulinum toxin injection for refractory overactive bladder treatment
- PMID: 40415074
- PMCID: PMC12104342
- DOI: 10.1038/s41598-025-03116-2
Ultrasound guided transabdominal botulinum toxin injection for refractory overactive bladder treatment
Abstract
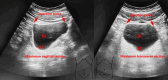
To evaluate the efficacy and safety of ultrasound - guided transabdominal injection of Botulinum toxin A (BoNT - A) in the treatment of refractory overactive bladder (rOAB). This retrospective cohort study included 64 patients with rOAB admitted to the Department of Urology, the Third People's Hospital of Hangzhou, from January 2021 to February 2025. They were divided into an observation group (ultrasound - guided transabdominal injection, 32 cases) and a control group (transurethral cystoscopic injection, 32 cases). Both groups received BoNT - A (100 U) intramural injection of the bladder. In the observation group, the injection was performed under real - time ultrasound guidance through the abdominal wall, while the control group underwent the standard cystoscopic injection method. The primary outcome measures included the parameters of the micturition diary (urgency episodes, daytime urination frequency, nocturia episodes) and bladder capacity (initial desire capacity, maximum bladder capacity) before treatment, 1 month, and 6 months post-treatment. The secondary outcomes covered the standardized scale scores of patients (ICIQ - OAB, OAB - Q), global impression of improvement (PGI - I), visual analog pain score (VAS), incidence of complications, retreatment rates, and immediate willingness to repeat the procedure. There were no statistically significant differences in baseline characteristics (age, sex, and pre - treatment symptom scores) between the two groups (all P > 0.05). After treatment, both groups of patients showed significant improvement in urgency episodes, daytime urination frequency, nocturia episodes, bladder capacity, and ICIQ - OAB and OAB - Q scores (all P < 0.05), but there was no significant difference in therapeutic effects between the groups (all P > 0.05).Six months after treatment, the observed indicators in the observation and control groups increased compared with 1 month after treatment, but were still significantly better than the baseline level (all P < 0.05). There was no significant difference in efficacy between the two groups (all P > 0.05). The incidence of complications in the observation group was significantly lower than that in the control group (P < 0.01), and the VAS pain score was lower (3.32 ± 1.25 vs. 4.82 ± 1.61, P = 0.006), with a higher immediate willingness to repeat the procedure (8.28 ± 1.54 vs. 6.86 ± 2.19, P = 0.004). There was no significant difference in PGI-I scores between the groups (2.27 ± 0.92 vs. 1.95 ± 0.43, P = 0.08). There was no significant difference in the repeat injection rate between the two groups at 6 months after treatment (28.13% vs. 18.75%, P = 0.375). Ultrasound - guided transabdominal injection of BoNT - A for rOAB is as effective as transurethral cystoscopic injection, but it can significantly reduce the risk of complications and improve patient acceptance of treatment, providing a safer and more compliant alternative option for clinical practice.
Keywords: Botulinum toxin type A; Micturition diary; Refractory overactive bladder; Ultrasound guidance; Urodynamics.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethical statement: This retrospective study was conducted in accordance with the ethical standards of the institutional and/or national research committee and with the Declaration of Helsinki.This study was conducted with approval from the Ethics Committee of Hangzhou Third People’s Hospital (Y-KL2025025). Written informed consent was obtained from all participants.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

