Characterization and neurogenic responses of primary and immortalized Müller glia
- PMID: 40417181
- PMCID: PMC12098385
- DOI: 10.3389/fcell.2025.1513163
Characterization and neurogenic responses of primary and immortalized Müller glia
Abstract
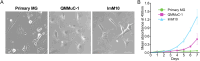
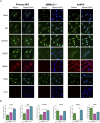
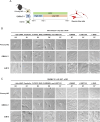
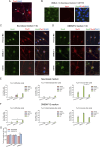
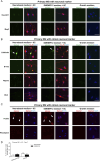
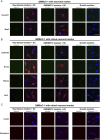
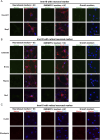
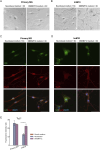
Primary Müller glia (MG) have been reported to exhibit a neurogenic capacity induced by small molecules. However, whether immortalized mouse MG cell lines exhibit neurogenic capacities similar to those of primary mouse MG remains unclear. In this study, we examined the morphology, proliferation rate, and marker profile of primary MG cells isolated from postnatal mouse pups with two immortalized mouse MG cell lines, QMMuC-1 and ImM10, in a standard growth medium. After chemical induction, we compared the morphology, markers, direct neuronal reprogramming efficiency, and axon length of these cell types in two culture media: Neurobasal and DMEM/F12. Our results showed that in standard growth medium, QMMuC-1 and ImM10 cells displayed similar morphology and marker profiles as primary MG cells, with the only differences observed in nestin expression. However, QMMuC-1 and ImM10 cells exhibited much higher proliferation rates than the primary MG cells. Following chemical treatment in both Neurobasal and DMEM/F12 media, a subset of primary MG, QMMuC-1, and ImM10 cells was induced to differentiate into immature neuron-like cells by day 7. While primary MG cells showed similar neuronal reprogramming efficiency and axon length extension in both media, QMMuC-1 and ImM10 cells displayed variations between the two culture media. Moreover, some of the induced neuronal cells derived from primary MG cells expressed HuC/D and Calbindin markers, whereas none of the cells derived from QMMuC-1 and ImM10 cells expressed these markers. Subsequent observations revealed that induced immature neuron-like cells derived from primary MG cells in both types of media and those derived from ImM10 cells cultured in DMEM/F12 survived until day 14. Taken together, our findings suggest that the two immortalized cell lines, QMMuC-1 and ImM10, exhibited neurogenic capacities similar to those of primary MG cells to some extent but did not fully recapitulate all their characteristics. Therefore, careful consideration should be given to culture conditions and the validation of key results when using immortalized cells as a substitute for primary MG cells.
Keywords: ImM10 cell line; Müller glia; QMMuC-1 cell line; chemical induction; neurogenic responses; neuronal reprogramming.
Copyright © 2025 Tran, Lukmanto, Chen, Strauß, Yamashita, Ohneda and Fukuda.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Characterization of a Spontaneously Immortalized Murine Müller Glial Cell Line QMMuC-1.Invest Ophthalmol Vis Sci. 2018 Mar 1;59(3):1666-1674. doi: 10.1167/iovs.17-23293. Invest Ophthalmol Vis Sci. 2018. PMID: 29625493
-
A conditional immortalized mouse muller glial cell line expressing glial and retinal stem cell genes.Invest Ophthalmol Vis Sci. 2010 Nov;51(11):5991-6000. doi: 10.1167/iovs.10-5395. Epub 2010 May 26. Invest Ophthalmol Vis Sci. 2010. PMID: 20505190 Free PMC article.
-
Comparison of Rat Primary Midbrain Neurons Cultured in DMEM/F12 and Neurobasal Mediums.Basic Clin Neurosci. 2021 Mar-Apr;12(2):205-212. doi: 10.32598/bcn.12.2.1568.1. Epub 2021 Mar 1. Basic Clin Neurosci. 2021. PMID: 34925717 Free PMC article.
-
Advances in the study of Müller glia reprogramming in mammals.Front Cell Neurosci. 2023 Dec 14;17:1305896. doi: 10.3389/fncel.2023.1305896. eCollection 2023. Front Cell Neurosci. 2023. PMID: 38155865 Free PMC article. Review.
-
Regulating the formation of Müller glia-derived progenitor cells in the retina.Glia. 2025 Jan;73(1):4-24. doi: 10.1002/glia.24635. Epub 2024 Oct 24. Glia. 2025. PMID: 39448874 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

