The Effects of Neuronal Fyn Knockdown in the Hippocampus in the Rat Kainate Model of Temporal Lobe Epilepsy
- PMID: 40422246
- PMCID: PMC12110385
- DOI: 10.3390/cells14100743
The Effects of Neuronal Fyn Knockdown in the Hippocampus in the Rat Kainate Model of Temporal Lobe Epilepsy
Abstract
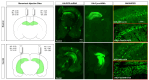
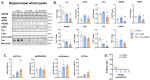
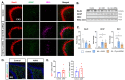
Previous studies have demonstrated neuronal and microglial Fyn, a Src family kinase (SFK), and how its interactions with tau contribute to epileptogenesis. Saracatinib, a Fyn/SFK inhibitor, modifies disease progression in rat kainate (KA) epilepsy models. In this study, we investigated neuronal-specific fyn knockdown effects on Fyn-tau signaling, neurodegeneration, and gliosis using a calcium/calmodulin-dependent protein kinase II (CaMKII)-promoter-driven adeno-associated viral vector (AAV9)-mediated fyn-shRNA injection in the rat hippocampus. Eight days following AAV administration, rats received repeated low-dose KA injections intraperitoneally to induce status epilepticus (SE). Both fyn-shRNA and control groups showed comparable SE severity, indicating inadequate neuronal fyn knockdown at this timepoint. Two weeks post fyn-shRNA injection, hippocampal Fyn significantly decreased, alongside reductions in NR2B, pNR2BY1472, PSD95, and total tau. There was also a compensatory activation of SFK (pSFKY416:Fyn) and tau hyperphosphorylation (AT8:total tau), negatively correlating with NeuN expression. Proximity ligation assay indicated unchanged Fyn-tau interactions, suggesting tau interactions with alternative SH3 domain proteins. Persistent neuronal loss, astrogliosis, and microgliosis suggested limited effectiveness of neuronal-specific fyn knockdown at this timepoint. An extended-duration fyn knockdown study, or using broad SFK inhibitors such as saracatinib or tau-SH3 blocking peptides, may effectively prevent SE-induced epileptogenesis.
Keywords: Src family kinase; epilepsy; fyn knockdown; neurodegeneration; neuroinflammation.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Figures


References
-
- CDC Epilepsy Basics. [(accessed on 15 March 2025)]; Available online: https://www.cdc.gov/epilepsy/about/index.html.
-
- Epilepsy. [(accessed on 4 April 2025)]. Available online: https://www.who.int/news-room/fact-sheets/detail/epilepsy.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

