Staphylococcus aureus Endocarditis Immunothrombosis
- PMID: 40422904
- PMCID: PMC12114098
- DOI: 10.3390/metabo15050328
Staphylococcus aureus Endocarditis Immunothrombosis
Abstract
Background: Infective endocarditis continues to represent a challenge for healthcare systems, requiring careful management and resources. Recent studies have indicated a shift in the predominant pathogens of concern, with Streptococcus sp. a being superseded by Staphylococcus sp. and Enterococcus sp. as the leading causes of concern. This shift is of concern as it is associated with Staphylococcus Aureus which has a high virulence rate and a tendency to form a biofilm, meaning that non-surgical therapy may not be effective. It is imperative to deliberate on the likelihood of platelet blood clot formation, which may be accompanied by bacterial infestation and the development of a biofilm. Methods: MEDLINE, Embase, and Pubmed were searched using terms relating to 'endocarditis' and 'Staphilococcus aureus', along with 'epidemiology', 'pathogenesis', 'coagulation', 'platelet', 'aggregation', and 'immunity'. The search focused on publications from the past 15 years, but excluded older, highly regarded articles. We also searched the reference lists of relevant articles. Recommended review articles are cited for more details. Results: An endocarditis lesion is believed to be a blood clot infected with bacteria that adheres to the heart valves. Infective endocarditis is a good example of immunothrombosis, where the coagulation system, innate immunity and the function of coagulation in isolating and eliminating pathogens interact. However, in the context of infective endocarditis, immunothrombosis unintentionally establishes an environment conducive to bacterial proliferation. The process of immunothrombosis impedes the immune system, enabling bacterial proliferation. The coagulation system plays a pivotal role in the progression of this condition. Conclusion: The coagulation system is key to how bacteria attach to the heart valves, how vegetations develop, and how complications like embolisation and valve dysfunction occur. Staphylococcus aureus, the main cause of infective endocarditis, can change blood clotting, growing well in the fibrin-rich environment of vegetation. The coagulation system is a good target for treating infective endocarditis because of its central role in the disease. But we must be careful, as using blood-thinning medicines in patients with endocarditis can often lead to an increased risk of bleeding.
Keywords: Staphylococcus aureus; Staphylococcus aureus infection; immunoresponse; infective endocarditis; platelet activation; thromboimmunity.
Conflict of interest statement
The author declares no conflict of interest.
Figures

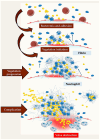


Similar articles
-
Coagulation: At the heart of infective endocarditis.J Thromb Haemost. 2020 May;18(5):995-1008. doi: 10.1111/jth.14736. Epub 2020 Feb 3. J Thromb Haemost. 2020. PMID: 31925863 Review.
-
Neutrophils Protect Against Staphylococcus aureus Endocarditis Progression Independent of Extracellular Trap Release.Arterioscler Thromb Vasc Biol. 2023 Feb;43(2):267-285. doi: 10.1161/ATVBAHA.122.317800. Epub 2022 Dec 1. Arterioscler Thromb Vasc Biol. 2023. PMID: 36453281 Free PMC article.
-
Isolated Pulmonary Valve Infective Endocarditis With Persistent Staphylococcus aureus Bacteremia and Rapid Clearance With Ertapenem Plus Cefazolin.Cureus. 2025 Jan 12;17(1):e77335. doi: 10.7759/cureus.77335. eCollection 2025 Jan. Cureus. 2025. PMID: 39935929 Free PMC article.
-
Type IV Pili of Streptococcus sanguinis Contribute to Pathogenesis in Experimental Infective Endocarditis.Microbiol Spectr. 2021 Dec 22;9(3):e0175221. doi: 10.1128/Spectrum.01752-21. Epub 2021 Nov 10. Microbiol Spectr. 2021. PMID: 34756087 Free PMC article.
-
Aspirin or Ticagrelor in Staphylococcus aureus Infective Endocarditis: Where Do We Stand?Front Cell Dev Biol. 2021 Oct 7;9:716302. doi: 10.3389/fcell.2021.716302. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34692677 Free PMC article. Review.
References
-
- Selton-Suty C., Célard M., Le Moing V., Doco-Lecompte T., Chirouze C., Iung B., Strady C., Revest M., Vandenesch F., Bouvet A., et al. Preeminence of Staphylococcus aureus in infective endocarditis: A 1-year population-based survey. Clin. Infect. Dis. 2012;54:1230–1239. doi: 10.1093/cid/cis199. - DOI - PubMed
-
- Murdoch D.R., Corey G.R., Hoen B., Miró J.M., Fowler V.G., Bayer A.S., Karchmer A.W., Olaison L., Pappas P.A., Moreillon P., et al. Clinical presentation, etiology, and outcome of infective endocarditis in the 21st century: The International Collaboration on Endocarditis-Prospective Cohort Study. Arch. Intern. Med. 2009;169:463–473. doi: 10.1001/archinternmed.2008.603. - DOI - PMC - PubMed
-
- Liaqat W., Palaiodimos L., Li W., Karamanis D., Tahir A., Tzoumas A., Nagraj S., Tiwari N., Grushko M., Kokkinidis D., et al. Epidemiologic and clinical characteristics of infective endocarditis: A single-center retrospective study in the Bronx, New York. Infection. 2022;50:1349–1361. doi: 10.1007/s15010-022-01846-3. - DOI - PubMed
-
- Delgado V., Ajmone Marsan N., de Waha S., Bonaros N., Brida M., Burri H., Caselli S., Doenst T., Ederhy S., Erba P.A., et al. 2023 ESC Guidelines for the management of endocarditis. Eur. Heart J. 2023;44:3948–4042. - PubMed
-
- Fowler V.G., Durack D.T., Selton-Suty C., Athan E., Bayer A.S., Chamis A.L., Dahl A., Di Bernardo L., Durante-Mangoni E., Duval X., et al. The 2023 Duke-International Society for Cardiovascular Infectious Diseases Criteria for Infective Endocarditis: Updating the Modified Duke Criteria. Clin. Infect. Dis. 2023;77:518–526. doi: 10.1093/cid/ciad271. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

