Prevalence of Actionable Exposures to Pharmacogenetic Medications Among Solid Organ Transplant Recipients in a Population-Scale Biobank
- PMID: 40423057
- PMCID: PMC12113073
- DOI: 10.3390/jpm15050185
Prevalence of Actionable Exposures to Pharmacogenetic Medications Among Solid Organ Transplant Recipients in a Population-Scale Biobank
Abstract
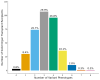
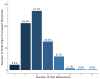
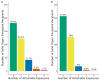
Background/Objectives: Solid organ transplant (SOT) recipients are exposed to multiple medications, many of which have pharmacogenetic (PGx) prescribing recommendations. This study leveraged data from a population-scale biobank and an enterprise data warehouse to determine the prevalence of actionable exposures to PGx medications among kidney, heart, and lung transplant recipients during the first six months post-transplant. Methods: We conducted a retrospective analysis of adult SOT patients with genetic data available from the Colorado Center for Personalized Medicine (CCPM) biobank and clinical data from Health Data Compass (HDC). We evaluated 29 variants in 13 pharmacogenes and 42 Clinical Pharmacogenetics Implementation Consortium (CPIC) level A or B medications (i.e., sufficient evidence to recommend at least one prescribing action based on genetics). The primary outcome was actionable exposure to a PGx medication (i.e., actionable phenotype and a prescription for an affected PGx medication). Results: The study included 358 patients. All patients were prescribed at least one PGx medication, and 49.4% had at least one actionable exposure to a PGx medication during the first six months post-transplant. The frequency of actionable exposure was highest for tacrolimus (15.4%), followed by proton pump inhibitors (PPIs) (15.1%) and statins (12.8%). Statin actionable exposures significantly differed by transplant type, likely due to variations in prescribing patterns and actionable phenotypes for individual statins. Conclusions: Our findings highlight the potential clinical utility of PGx testing among SOT patients. Further studies are needed to address the impact on clinical outcomes and the optimal timing of PGx testing in the SOT population.
Keywords: biobank; pharmacogenetics; pharmacogenomics; solid organ transplant.
Conflict of interest statement
K.M.D. was an employee of the University of Colorado at the time of this work and is currently an employee of Amgen Pharmaceuticals. All other authors report no conflicts of interest.
Figures
Similar articles
-
Evaluating the frequency and the impact of pharmacogenetic alleles in an ancestrally diverse Biobank population.J Transl Med. 2022 Nov 28;20(1):550. doi: 10.1186/s12967-022-03745-5. J Transl Med. 2022. PMID: 36443877 Free PMC article.
-
Prescription Patterns and Relationship to Pharmacogenomics Testing in the Military Health System.Mil Med. 2021 Dec 30;187(Suppl 1):9-17. doi: 10.1093/milmed/usab481. Mil Med. 2021. PMID: 34967405
-
Impact of Pharmacogenomic Testing in Pediatric Heart and Kidney Transplant.Pediatr Transplant. 2025 Mar;29(2):e70044. doi: 10.1111/petr.70044. Pediatr Transplant. 2025. PMID: 39924350
-
Barriers to Implementing Clinical Pharmacogenetics Testing in Sub-Saharan Africa. A Critical Review.Pharmaceutics. 2020 Aug 26;12(9):809. doi: 10.3390/pharmaceutics12090809. Pharmaceutics. 2020. PMID: 32858798 Free PMC article. Review.
-
Pharmacogenetic Panel Testing: A Review of Current Practice and Potential for Clinical Implementation.Annu Rev Pharmacol Toxicol. 2025 Jan;65(1):91-109. doi: 10.1146/annurev-pharmtox-061724-080935. Epub 2024 Dec 17. Annu Rev Pharmacol Toxicol. 2025. PMID: 39348848 Review.
References
-
- Singh T.P., Cherikh W.S., Hsich E., Lewis A., Perch M., Kian S., Hayes D., Jr., Potena L., Stehlik J., Zuckermann A., et al. Graft survival in primary thoracic organ transplant recipients: A special report from the International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation. J. Heart Lung Transplant. 2023;42:1321–1333. doi: 10.1016/j.healun.2023.07.017. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

