Semaglultide targets Spp1+ microglia/macrophage to attenuate neuroinflammation following perioperative stroke
- PMID: 40426210
- PMCID: PMC12117698
- DOI: 10.1186/s12974-025-03465-9
Semaglultide targets Spp1+ microglia/macrophage to attenuate neuroinflammation following perioperative stroke
Abstract
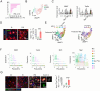
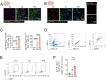
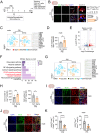
Peripheral surgery evokes neuroimmune activation in the central nervous system and modulates immune cell polarization in the ischemic brain. However, the phenotypic change of microglia and myeloid cells within post-surgical ischemic brain tissue remain poorly defined. Using an integrated approach that combines single-cell RNA sequencing with comprehensive biological analysis in a perioperative ischemic stroke (PIS) model, we identified a distinct Spp1-positive macrophage/microglia (Spp1+ Mac/MG) subgroup that exhibit enriched anti-inflammatory pathways with distinct lipid metabolic reprogrammed profile. Moreover, using immunofluorescence staining, we identified the expression of Glucagon-like peptide-1 receptor (GLP1R) in Spp1+F4/80+ cells and Spp1+Iba-1+ cells. Intraperitoneal administration of semaglutide, a GLP1R agonist clinically approved for the treatment of type 2 diabetes mellitus, resulted in a significant reduction of cerebral infarct volume in PIS mice compared to that in ischemic stroke (IS) mice. Meanwhile, semaglutide treatment also increased the proportion of Spp1+Edu+Iba-1+ cells 3 days after PIS. Using high-parameter flow cytometry, immunofluorescence staining and RNA sequencing, we demonstrated that semaglutide treatment significantly attenuated the expression of neuroinflammatory markers in mice following PIS. We also found that semaglutide treatment significantly ameliorated sensorimotor dysfunction up to 3 days after PIS in mice. Our current finding reveal a novel protective Spp1+Mac/MG subset after PIS and demonstrated that it can be upregulated by semaglutide. We propose that targeting Spp1+Mac/MG subsets using semaglutide could serve as a promising strategy to attenuate the exacerbated neuroinflammation in PIS.
Keywords: Neuroinflammation; Perioperative ischemic stroke; Semaglutide; Spp1+ Mac/MG.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethical approval: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures




References
-
- Gelderblom M, et al. Temporal and Spatial dynamics of cerebral immune cell accumulation in stroke. Stroke. 2009;40(5):1849–57. - PubMed
-
- Zhou L, et al. Treatment of cerebral ischemia by disrupting ischemia-induced interaction of nNOS with PSD-95. Nat Med. 2010;16(12):1439–43. - PubMed
-
- Pocock JM, Piers TM. Modelling microglial function with induced pluripotent stem cells: an update. Nat Rev Neurosci. 2018;19(8):445–52. - PubMed
MeSH terms
Substances
Grants and funding
- 82401524/the National Natural Science Foundation of China
- U22A20295/the National Natural Science Foundation of China
- 24ZR1444700/Shanghai "Science and Technology Innovation Action Plan" Natural Science Foundation
- No. 24X010202059/the Fundamental Research Funds for the Central Universities
- No. 20DZ2254200/Shanghai Engineering Research Center of Peri-operative Organ Support and Function Preservation
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

