The Size and Localization of Ribeye and GluR2 in the Auditory Inner Hair Cell Synapse of C57BL/6 Mice Are Affected by Short-Pulse Corticosterone in a Sex-Dependent Manner
- PMID: 40426612
- PMCID: PMC12110336
- DOI: 10.3390/brainsci15050441
The Size and Localization of Ribeye and GluR2 in the Auditory Inner Hair Cell Synapse of C57BL/6 Mice Are Affected by Short-Pulse Corticosterone in a Sex-Dependent Manner
Abstract
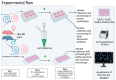
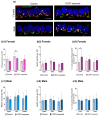
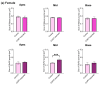
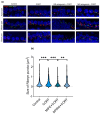
Background: Inner hair cell (IHC) ribbon synapses are the initial synapses in the auditory pathway, comprising presynaptic ribbons and postsynaptic glutamate receptors on the peripheral afferent fibers. The excitatory neurotransmitter glutamate primarily signals through AMPA-type heterotetrameric receptors (AMPARs), composed of GluR1, GluR2, GluR3, and GluR4 subunits. Research shows that corticosterone affects AMPA receptor subunits in the central nervous system. The present study investigates the effects of corticosterone on AMPA receptor subunits in the murine cochlea. Methods: Cochlear explants were isolated from male and female C57BL/6 pups (postnatal days 4-5), treated for 20 min with 100 nM corticosterone, and cultured for an additional 24 h. The concentration of AMPAR protein subunits was quantified using an ELISA assay, while gene expression was analyzed using RT-PCR. The synaptic localization patterns of GluR2 and Ribeye were examined using immunofluorescence and confocal microscopy. Results: Male C57BL/6 mice have a significantly greater basal concentration of the GluR2 subunit than females and more GluR2 puncta per IHC than females. Corticosterone increases the size of Ribeye in males and increases twofold GluR2/Ribeye colocalization in the apical region of females. Conclusions: Exposure of membranous cochleae to corticosterone induces changes consistent with neuroplasticity in the auditory periphery. The observed effect is sex-dependent.
Keywords: AMPA; cochlea; corticosterone; glutamate receptor subunits; inner hair cell synapse; stress hormones.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Widespread expression of the AMPA receptor GluR2 subunit at glutamatergic synapses in the rat spinal cord and phosphorylation of GluR1 in response to noxious stimulation revealed with an antigen-unmasking method.J Neurosci. 2004 Jun 23;24(25):5766-77. doi: 10.1523/JNEUROSCI.1237-04.2004. J Neurosci. 2004. PMID: 15215299 Free PMC article.
-
Loss of inner hair cell ribbon synapses and auditory nerve fiber regression in Cldn14 knockout mice.Hear Res. 2020 Jun;391:107950. doi: 10.1016/j.heares.2020.107950. Epub 2020 Mar 16. Hear Res. 2020. PMID: 32251970
-
Neurokinin 1 receptor-expressing projection neurons in laminae III and IV of the rat spinal cord have synaptic AMPA receptors that contain GluR2, GluR3 and GluR4 subunits.Eur J Neurosci. 2009 Feb;29(4):718-26. doi: 10.1111/j.1460-9568.2009.06633.x. Epub 2009 Feb 6. Eur J Neurosci. 2009. PMID: 19200070 Free PMC article.
-
A Scoping Review of Corticosterone-Induced Changes in Ionotropic Glutamate Receptor Levels and Localization in the Rodent Brain: Implications for the Auditory System.Brain Sci. 2025 Jan 24;15(2):110. doi: 10.3390/brainsci15020110. Brain Sci. 2025. PMID: 40002443 Free PMC article. Review.
-
The AMPAR subunit GluR2: still front and center-stage.Brain Res. 2000 Dec 15;886(1-2):190-207. doi: 10.1016/s0006-8993(00)02951-6. Brain Res. 2000. PMID: 11119696 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

