Investigation of the Effect of Amniomax on Lidocaine-Induced Toxicity in Healthy Colon Cell Culture
- PMID: 40426902
- PMCID: PMC12108911
- DOI: 10.3390/biomedicines13051074
Investigation of the Effect of Amniomax on Lidocaine-Induced Toxicity in Healthy Colon Cell Culture
Abstract
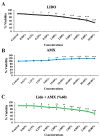
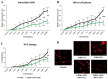
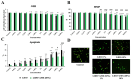
Background: Lidocaine (LIDO) toxicity is a critical concern in regional anesthesia, with no specific antidote currently available. While lipid emulsions are commonly used as rescue agents in cases of local anesthetic systemic toxicity (LAST), their efficacy is inconsistent, and their safety remains controversial. AmnioMax® (AMX), a specialized cell culture medium enriched with growth factors and bioactive molecules, has the potential to offer cytoprotective effects. This study aims to investigate the therapeutic efficacy of AMX in mitigating lidocaine-induced cytotoxicity and to explore its protective mechanisms at the cellular level. Methods: Healthy colon cells (CCD-18Co) were used in this study. Four experimental groups were established as follows: control, LIDO, AMX, and LIDO + AMX. Cellular viability in the control group was set at 100%. LIDO was administered at concentrations ranging from 0.06 to 10%, AMX at 0.625-100%, and LIDO + AMX at 60% of the half-maximal effective concentration (EC50) combined with LIDO (0.06-10%). Cells were incubated for 24 h, after which cellular viability, DNA damage, apoptosis, intracellular reactive oxygen species (iROS), intracellular calcium (Ca), mitochondrial membrane potential (MMP), and glutathione (GSH) were evaluated. Results: LIDO exposure led to a concentration-dependent decrease in viability compared to the control group (p < 0.001), while AMX significantly increased viability (p < 0.001). In the LIDO + AMX group, viability was also reduced (p < 0.001); however, cytotoxicity was significantly lower than in the LIDO group (p < 0.05). Both the LIDO and LIDO + AMX groups showed increased iROS levels, DNA damage, and apoptosis (p < 0.001), along with the decreased MMP and GSH levels (p < 0.001) compared to the control. However, in the LIDO + AMX group, iROS, DNA damage, and apoptosis were significantly lower than in the LIDO group (p < 0.01), MMP levels were increased (p < 0.001), and no significant difference was observed in GSH levels. Conclusions: AMX demonstrated cytoprotective effects against LIDO-induced cytotoxicity, suggesting its potential as an alternative therapeutic agent for LAST.
Keywords: AmnioMax®; apoptosis; cell culture; cytotoxicity; lidocaine; viability.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Inhibitory gene expression of the Cav3.1 T-type calcium channel to improve neuronal injury induced by lidocaine hydrochloride.Eur J Pharmacol. 2016 Mar 15;775:43-9. doi: 10.1016/j.ejphar.2016.02.019. Epub 2016 Feb 4. Eur J Pharmacol. 2016. PMID: 26852957
-
Investigation of the Protective Effects of Magnesium on Bupivacaine-Induced Toxicity at the Level of Colon Cell Culture.Biomedicines. 2024 Jul 24;12(8):1652. doi: 10.3390/biomedicines12081652. Biomedicines. 2024. PMID: 39200118 Free PMC article.
-
Lidocaine alleviates cytotoxicity-resistance in lung cancer A549/DDP cells via down-regulation of miR-21.Mol Cell Biochem. 2019 Jun;456(1-2):63-72. doi: 10.1007/s11010-018-3490-x. Epub 2019 Jan 14. Mol Cell Biochem. 2019. Retraction in: Mol Cell Biochem. 2024 Mar;479(3):737. doi: 10.1007/s11010-024-04971-9. PMID: 30644017 Retracted.
-
Lido-OH, a Hydroxyl Derivative of Lidocaine, Produced a Similar Local Anesthesia Profile as Lidocaine With Reduced Systemic Toxicities.Front Pharmacol. 2021 Sep 16;12:678437. doi: 10.3389/fphar.2021.678437. eCollection 2021. Front Pharmacol. 2021. PMID: 34603015 Free PMC article.
-
Dietary glycation compounds - implications for human health.Crit Rev Toxicol. 2024 Sep;54(8):485-617. doi: 10.1080/10408444.2024.2362985. Epub 2024 Aug 16. Crit Rev Toxicol. 2024. PMID: 39150724
References
-
- Yasmeen S., Liao X., Khan F.U., Ihsan A.U., Li X., Li C., Chen D., Yu F., Wang Z., Sembatya K.R., et al. A novel approach to devise the therapy for ventricular fibrillation by epicardial delivery of lidocaine using active hydraulic ventricular attaching support system: An experimental study in rats. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019;107:1722–1731. doi: 10.1002/jbm.b.34265. - DOI - PubMed
-
- Mirshahidi S., Shields T.G., de Necochea-Campion R., Yuan X., Janjua A.B., Williams N.L., Mirshahidi H.R., Reeves M.E., Duerksen-Hughes P., Zuckerman L.M. Bupivacaine and Lidocaine Induce Apoptosis in Osteosarcoma Tumor Cells. Clin. Orthop. Relat. Res. 2021;479:180–194. doi: 10.1097/CORR.0000000000001510. - DOI - PMC - PubMed
-
- Neal J.M., Barrington M.J., Fettiplace M.R., Gitman M., Memtsoudis S.G., Mörwald E.E., Rubin D.S., Weinberg G. The Third American Society of Regional Anesthesia and Pain Medicine Practice Advisory on Local Anesthetic Systemic Toxicity: Executive Summary 2017. Reg. Anesth. Pain Med. 2018;43:113–123. doi: 10.1097/AAP.0000000000000720. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

