Targeting Metabolic Reprogramming in Bladder Cancer Immunotherapy: A Precision Medicine Approach
- PMID: 40426972
- PMCID: PMC12108893
- DOI: 10.3390/biomedicines13051145
Targeting Metabolic Reprogramming in Bladder Cancer Immunotherapy: A Precision Medicine Approach
Abstract
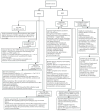
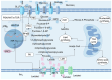
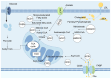
Bladder cancer, as a highly heterogeneous malignant tumor of the urinary system, is significantly affected by tumor metabolic reprogramming in its response to immunotherapy. This review systematically elaborates on the molecular mechanisms of abnormal glucose and lipid metabolism in the bladder cancer microenvironment and immune escape, and discusses precision treatment strategies based on metabolic regulation. In the future, it will be necessary to combine spatiotemporal omics and artificial intelligence technologies to construct a multi-target intervention system for the metabolic-immune interaction network, promoting a paradigm shift in precision treatment for bladder cancer.
Keywords: bladder cancer; immunotherapy; metabolic reprogramming; tumor microenvironment.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Metabolic reprogramming and immunological changes in the microenvironment of esophageal cancer: future directions and prospects.Front Immunol. 2025 Jan 24;16:1524801. doi: 10.3389/fimmu.2025.1524801. eCollection 2025. Front Immunol. 2025. PMID: 39925801 Free PMC article. Review.
-
The role of metabolic reprogramming in immune escape of triple-negative breast cancer.Front Immunol. 2024 Aug 13;15:1424237. doi: 10.3389/fimmu.2024.1424237. eCollection 2024. Front Immunol. 2024. PMID: 39192979 Free PMC article. Review.
-
Emerging role of metabolic reprogramming in the immune microenvironment and immunotherapy of thyroid cancer.Int Immunopharmacol. 2025 Jan 10;144:113702. doi: 10.1016/j.intimp.2024.113702. Epub 2024 Nov 26. Int Immunopharmacol. 2025. PMID: 39602959 Review.
-
Metabolic reprogramming in tumor immune microenvironment: Impact on immune cell function and therapeutic implications.Cancer Lett. 2024 Aug 10;597:217076. doi: 10.1016/j.canlet.2024.217076. Epub 2024 Jun 19. Cancer Lett. 2024. PMID: 38906524 Review.
-
Involvement of tumor immune microenvironment metabolic reprogramming in colorectal cancer progression, immune escape, and response to immunotherapy.Front Immunol. 2024 Jul 25;15:1353787. doi: 10.3389/fimmu.2024.1353787. eCollection 2024. Front Immunol. 2024. PMID: 39119332 Free PMC article. Review.
Cited by
-
The impact of METTL3 on bladder cancer through m6A modification: a potential therapeutic target and prognostic biomarker.Front Oncol. 2025 Jul 3;15:1622117. doi: 10.3389/fonc.2025.1622117. eCollection 2025. Front Oncol. 2025. PMID: 40678060 Free PMC article. Review.
References
-
- Smani S., DuBois J., Zhao K., Sutherland R., Rahman S.N., Humphrey P., Hesse D., Tan W.S., Martin D., Lokeshwar S.D., et al. Advancements in the Diagnosis, Treatment, and Risk Stratification of Non-Muscle Invasive Bladder Cancer. Curr. Oncol. Rep. 2025;27:236–246. doi: 10.1007/s11912-025-01645-7. - DOI - PubMed
-
- Netto G.J., Amin M.B., Berney D.M., Compérat E.M., Gill A.J., Hartmann A., Menon S., Raspollini M.R., Rubin M.A., Srigley J.R., et al. The 2022 World Health Organization Classification of Tumors of the Urinary System and Male Genital Organs-Part B: Prostate and Urinary Tract Tumors. Eur. Urol. 2022;82:469–482. doi: 10.1016/j.eururo.2022.07.002. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

