Exploring Protein Misfolding in Amyotrophic Lateral Sclerosis: Structural and Functional Insights
- PMID: 40426973
- PMCID: PMC12109280
- DOI: 10.3390/biomedicines13051146
Exploring Protein Misfolding in Amyotrophic Lateral Sclerosis: Structural and Functional Insights
Abstract
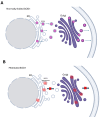
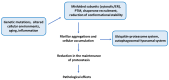
Protein functionality depends on its proper folding, making protein misfolding crucial for the function of proteins and, by extension, cells and the whole organism. Increasing evidence supports the role of protein misfolding in the pathogenesis of neurodegenerative diseases, such as amyotrophic lateral sclerosis (ALS). ALS is a rapidly progressive disease diagnosed at a prevalence of 5 cases per 100,000, with approximately 2-3 patients per 100,000 diagnosed each year. To date, there is no cure, and the disease usually leads to death within 2 to 5 years from diagnosis. There are two types of the disorder: familial ALS (fALS), accounting for approximately 10% of cases, and sporadic (sALS), accounting for the remaining 90%. The hallmark of ALS, regardless of type, is the protein aggregates found in patients' tissues. This suggests that the disruption of proteostasis plays a critical role in the development of the disease. Herein, we stress the distinct factors that lead to protein misfolding and aggregate formation in ALS. Specifically, we highlight several triggering factors affecting protein misfolding, namely mutations, errors in the processes of protein production and trafficking, and failures of folding and chaperone machinery. Gaining a deeper understanding of protein aggregation will improve our comprehension of disease pathogenesis and potentially uncover new therapeutic approaches.
Keywords: aggregation; amyotrophic lateral sclerosis; mutations; prion; protein misfolding; superoxide dismutase 1.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Devi S., Chaturvedi M., Fatima S., Priya S. Environmental factors modulating protein conformations and their role in protein aggregation diseases. Toxicology. 2022;465:153049. - PubMed
-
- Khan A.N., Khan R.H. Protein misfolding and related human diseases: A comprehensive review of toxicity, proteins involved, and current therapeutic strategies. Int. J. Biol. Macromol. 2022;223 Pt A:143–160. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

