Recycled Sericin Biopolymer in Biotechnology and Bioelectronics
- PMID: 40428166
- PMCID: PMC12108825
- DOI: 10.3390/bioengineering12050547
Recycled Sericin Biopolymer in Biotechnology and Bioelectronics
Abstract
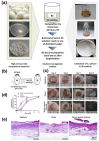
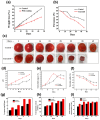
In a world characterized by rapid industrialization and a growing population, plastic or polymeric waste handling has undergone significant transformations. Recycling has become a major strategy where silk sericin has great potential among recyclable polymers. This naturally occurring biopolymer is a sustainable and versatile material with a wide range of potential uses in biotechnology and sensing. Furthermore, preparing and studying new environmentally friendly functional polymers with attractive physicochemical properties can open new opportunities for developing next-generation materials and composites. Herein, we provide an overview of the advances in the research studies of silk sericin as a functional and eco-friendly material, considering its biocompatibility and unique physicochemical properties. The structure of silk sericin and the extraction procedures, considering the influence of preparation methods on its properties, are described. Sericin's intrinsic properties, including its ability to crosslink with other polymers, its antioxidative capacity, and its biocompatibility, render it a versatile material for multifunctional applications across diverse fields. In biotechnology, the ability to blend sericin with other polymers enables the preparation of materials with varied morphologies, such as films and scaffolds, exhibiting enhanced mechanical strength and anti-inflammatory effects. This combination proves particularly advantageous in tissue engineering and wound healing. Furthermore, the augmentation of mechanical strength, coupled with the incorporation of plasticizers, makes sericin films suitable for the development of epidermal electrodes. Simultaneously, by precisely controlling hydration and permeability, the same material can be tailored for applications in packaging and the food industry. This work highlights the multidisciplinary and multifunctional nature of sericin, emphasizing its broad applicability.
Keywords: bioelectronics; biopolymers; biosensors; biotechnology; green chemistry; silk sericin; sustainability; tissue engineering.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




References
-
- Bandh S.A., Malla F.A., Wani S.A., Hoang A.T. Waste Management in the Circular Economy. Springer; Cham, Switzerland: 2024. Waste Management and Circular Economy; pp. 1–17.
-
- Marques G.G., Couffin A., Hajji P., Inoubli R., Bounor-Legaré V., Fulchiron R. A Review on the Formulation and Rupture Properties of Polyethylene Terephthalate in a Mechanical Recycling Context. Ind. Eng. Chem. Res. 2023;63:887–920. doi: 10.1021/acs.iecr.3c02376. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

