The Effect of Lesion Length on Doppler Velocities Used Routinely to Determine Carotid Stenosis Cross-Sectional Severity
- PMID: 40428257
- PMCID: PMC12110091
- DOI: 10.3390/diagnostics15101259
The Effect of Lesion Length on Doppler Velocities Used Routinely to Determine Carotid Stenosis Cross-Sectional Severity
Abstract
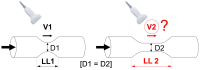
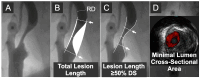
Background/Objective: Transcutaneous Doppler ultrasound is a fundamental tool in evaluating carotid stenosis cross-sectional severity (CS-CSS) in clinical practice because peak-systolic and end-diastolic velocities (PSV, EDV) increase with angiographic diameter stenosis. We tested the hypothesis that lesion length (LL) may affect PSV and EDV. Methods: CARUS (Carotid Artery IntravasculaR Ultrasound Study) prospectively enrolled 300 consecutive patients (age 47-83 years, 64.3% men, 63.3% symptomatic) with carotid stenosis ≥50% by Doppler ultrasound considered diagnostic (corelab analyst). We correlated stenosis LL (mm) and minimal lumen area (MLA, mm2) with PSV and EDV. Results: IVUS imaging (20 MHz Volcano/Philips) was uncomplicated. As IVUS probe forward/backward movement with systole/diastole ("jumping"-related artifact superimposed on motorized pullback) restrained LL (but not MLA) determination, LL measurement was angiographic. Final data set included 293 patients/stenoses (applicable to seven angiograms unsuitable for LL measurement). Irrespective of CS-CSS, a significant LL effect on PSV and EDV occurred with LL ≥ 7 mm (n = 224/293, i.e., 76.5% study patients/lesions; r = 0.38 and r = 0.35); for MLA irrespective of CS-CCS the coefficients were r = 0.49 (PSV) and r = 0.42 (EDV); p < 0.001 for all. For LL and MLA considered together, the correlations were stronger: r = 0.61 (PSV) and r = 0.54 (EDV); p < 0.0001 for both. Combined LL and MLA effect was represented by the following formulas: PSV = 0.31 × LL/MLA + 2.02 [m/s]; EDV = 0.12 × LL/MLA + 0.63 [m/s], enabling to correct the PSV (EDV)-based assessment of CS-CSS for the LL effect. Conclusions: This study provides, for the first time, systematic evidence that the length of carotid stenosis significantly affects lesional Doppler velocities. We established formulas incorporating the contribution of both stenosis length and its cross-sectional severity to PSV and EDV. We advocate including stenosis length measurement in duplex ultrasound reports when performing PSV (EDV)-based assessment of carotid cross-sectional stenosis severity.
Keywords: carotid stenosis; cross-sectional stenosis; doppler ultrasound; duplex ultrasound; lesion length; stenosis severity; stenotic segment length.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Imaging modality-dependent carotid stenosis severity variations against intravascular ultrasound as a reference: Carotid Artery intravasculaR Ultrasound Study (CARUS).Int J Cardiovasc Imaging. 2023 Oct;39(10):1909-1920. doi: 10.1007/s10554-023-02875-1. Epub 2023 Aug 21. Int J Cardiovasc Imaging. 2023. PMID: 37603155 Free PMC article.
-
Carotid duplex ultrasound velocity measurements versus intravascular ultrasound in detecting carotid in-stent restenosis.Circ Cardiovasc Interv. 2009 Oct;2(5):438-43. doi: 10.1161/CIRCINTERVENTIONS.109.857276. Epub 2009 Sep 1. Circ Cardiovasc Interv. 2009. PMID: 20031754
-
Validating common carotid stenosis by duplex ultrasound with carotid angiogram or computed tomography scan.J Vasc Surg. 2014 Feb;59(2):435-9. doi: 10.1016/j.jvs.2013.08.030. Epub 2013 Sep 29. J Vasc Surg. 2014. PMID: 24080127
-
Carotid artery stenosis: grayscale and Doppler ultrasound diagnosis--Society of Radiologists in Ultrasound consensus conference.Ultrasound Q. 2003 Dec;19(4):190-8. doi: 10.1097/00013644-200312000-00005. Ultrasound Q. 2003. PMID: 14730262 Review.
-
Joint recommendations for reporting carotid ultrasound investigations in the United Kingdom.Eur J Vasc Endovasc Surg. 2009 Mar;37(3):251-61. doi: 10.1016/j.ejvs.2008.10.015. Epub 2008 Nov 29. Eur J Vasc Endovasc Surg. 2009. PMID: 19046904 Review.
References
-
- Musialek P., Bonati L.H., Bulbulia R., Halliday A., Bock B., Capoccia L., Eckstein H.H., Grunwald I.Q., Lip P.L., Monteiro A., et al. Stroke risk management in carotid atherosclerotic disease: A Clinical Consensus Statement of the ESC Council on Stroke and the ESC Working Group on Aorta and Peripheral Vascular Diseases. Cardiovasc. Res. 2025;121:13–43. doi: 10.1093/cvr/cvad135. - DOI - PubMed
-
- Tekieli L., Dzierwa K., Grunwald I.Q., Mazurek A., Urbanczyk-Zawadzka M., Wiewiorka L., Banys R.P., Dabrowski W., Podlasek A., Weglarz E., et al. Outcomes in acute carotid-related stroke eligible for mechanical reperfusion: SAFEGUARD-STROKE Registry. J. Cardiovasc. Surg. 2024;65:231–248. doi: 10.23736/S0021-9509.24.13093-5. - DOI - PubMed
-
- Powers W.J., Rabinstein A.A., Ackerson T., Adeoye O.M., Bambakidis N.C., Becker K., Biller J., Brown M., Demaerschalk B.M., Hoh B., et al. Guidelines for the Early Management of Patients with Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50:e344–e418. - PubMed
-
- Tekieli L., Afanasjev A., Mazgaj M., Borodetsky V., Sievert K., Ruzsa Z., Knapik M., Širvinskas A., Mazurek A., Dzierwa K., et al. A multi-center study of the MicroNET-covered stent in consecutive patients with acute carotid-related stroke: SAFEGUARD-STROKE. Adv. Interv. Cardiol. 2024;20:172–193. doi: 10.5114/aic.2024.140963. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

