Modification of Natural and Synthetic Zeolites for CO2 Capture: Unrevealing the Role of the Compensation Cations
- PMID: 40429140
- PMCID: PMC12113214
- DOI: 10.3390/ma18102403
Modification of Natural and Synthetic Zeolites for CO2 Capture: Unrevealing the Role of the Compensation Cations
Abstract
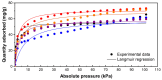
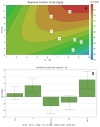
The development of highly effective natural-based adsorbents to face the increasing rates of CO2 production and their delivery to the atmosphere are a big concern nowadays. For such purposes, synthetic and natural zeolites were modified via an ion exchange procedure to enhance the CO2 uptake. Samples were characterized by SEM, EDS, TGA and nitrogen adsorption at 77 K, showing the correct incorporation of the new metals; in addition, the CO2 adsorption isotherms were determined using a gas analyser. During the first stage, the role of the compensation cations for CO2 adsorption was assessed by modifying a pure ZSM-5 synthetic zeolite with different metal precursors present in salt solutions via an ion exchange procedure. Then, five samples were studied; the samples modified with bivalent cation precursors (Zn2+ and Cu2+) presented a higher adsorption uptake than those modified with a monovalent cation (Na+ and K+). Specifically, the substitution of the compensation cations for Cu2+ increased the CO2 capture uptake without affecting the surface properties of the zeolite. The results depict the prevalence of π-cation interactions enhanced by the field gradient induced by divalent cations and their lower ionic radii, if compared to monovalent ones. Subsequently, a natural zeolite was modified considering the best results of the previous phase. This Surface Response Methodology was implemented considering 11 samples by varying the concentration of the copper precursor and the time of the ion exchange procedure. A quantitative quadratic model to predict the adsorption uptake with an R2 of 0.92 was obtained. The results depicted the optimal conditions to modify the used natural zeolite for CO2 capture. The modification procedure implemented increased the CO2 adsorption capacity of the natural zeolite more than 20%, reaching an adsorption capacity of 75.8 mg CO2/g zeolite.
Keywords: CO2 adsorption; carbon capture and storage; ion exchange; isotherm modelling; zeolite.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





References
-
- Wei C., Wang M., Fu Q., Dai C., Huang R., Bao Q. Temporal Characteristics of Greenhouse Gases (CO2 and CH4) in the Megacity Shanghai, China: Association with Air Pollutants and Meteorological Conditions. Atmos. Res. 2020;235:104759. doi: 10.1016/j.atmosres.2019.104759. - DOI
-
- Dinda S., Murge P., Chakravarthy Paruchuri B. A Study on Zeolite-Based Adsorbents for CO2 Capture. Bull. Mater. Sci. 2019;42:240. doi: 10.1007/s12034-019-1936-8. - DOI
-
- Olivier J.G.J., Peters J.A.H.W. 2020 Report Trends in Global CO2 and Total Greenhouse Gas Emissions: 2020 Report. PBL Netherlands Environmental Assessment Agency; Den Haag, The Netherlands: 2020. Trends in global CO2 and total greenhouse gas emissions.
-
- Intergovernmental Panel on Climate Change (IPCC) Climate Change 2022—Impacts, Adaptation and Vulnerability. Cambridge University Press; Singapore: 2023.
-
- Swiss Re Partnering for Progress Financial Highlights. [(accessed on 19 April 2025)]. Available online: https://www.swissre.com/dam/jcr:59cd3668-b9ef-4746-b9d3-132139c461a6/ar-....
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

