Perspective on Renal Involvement in Antiphospholipid Syndrome: Implications for Diagnosis, Pathogenesis, and Treatment
- PMID: 40429322
- PMCID: PMC12112115
- DOI: 10.3390/jcm14103326
Perspective on Renal Involvement in Antiphospholipid Syndrome: Implications for Diagnosis, Pathogenesis, and Treatment
Abstract
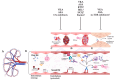
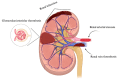
Antiphospholipid syndrome (APS) can affect the kidneys, leading to renal artery and vein thrombosis, allograft loss following transplantation, and microvascular damage referred to as aPL-nephropathy (aPL-N). APL-N is a complex and frequently underdiagnosed condition characterized by an incomplete understanding of its etiopathogenesis and associated with unfavorable renal outcomes. The 2023 ACR/EULAR classification criteria for APS included aPL-N within the microvascular domain. The gold standard for aPL-N is the biopsy, revealing lesions associated with acute thrombotic microangiopathy and chronic vascular changes. Nevertheless, reluctance for biopsies due to anticoagulation and thrombocytopenia underscores the need for noninvasive diagnostics. Common clinical features include hypertension, microscopic hematuria, proteinuria, and renal insufficiency. Antiphospholipid antibodies seem crucial to kidney damage through thrombotic and inflammatory processes. Studies and experimental models of thrombotic microangiopathy lesions suggest the involvement of the complement cascade, tissue factor, and mammalian target of the rapamycin complex activation pathway. Currently, the management of aPL-N is based mainly on expert opinion, with limited evidence supporting the use of anticoagulants, leading to controversy in their application. Treatment may include heparin, intravenous immunoglobulin, plasma exchange, and targeted therapies tailored to aPL-N mechanisms. Future multicenter studies are essential to clarify their roles. The goal of this review is to inform clinicians and create a research agenda to address the unmet needs in diagnosing and managing APL-N.
Keywords: aPL-nephropathy; antiphospholipid antibodies; lupus anticoagulant; thrombosis; thrombotic angiopathy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Miyakis S., Lockshin M.D., Atsumi T., Branch D.W., Brey R.L., Cervera R., Derksen R.H., DEGroot P.G., Koike T., Meroni P.L., et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS) J. Thromb. Haemost. 2006;4:295–306. doi: 10.1111/j.1538-7836.2006.01753.x. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

