Opposing Calcium-Dependent Effects of GsMTx4 in Acute Lymphoblastic Leukemia: In Vitro Proliferation vs. In Vivo Survival Advantage
- PMID: 40429963
- PMCID: PMC12112700
- DOI: 10.3390/ijms26104822
Opposing Calcium-Dependent Effects of GsMTx4 in Acute Lymphoblastic Leukemia: In Vitro Proliferation vs. In Vivo Survival Advantage
Abstract
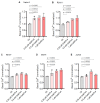
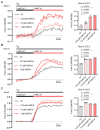
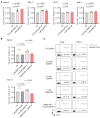
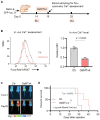
Mechanogated (MG) ion channels play a crucial role in mechano-transduction and immune cell regulation, yet their impact on blood cancers, particularly acute lymphoblastic leukemia (ALL), remains poorly understood. This study investigates the pharmacological effects of GsMTx4, an MG channel inhibitor, in human ALL cells both in vitro and in vivo. Unexpectedly, we found that GsMTx4 remarkably increased basal calcium (Ca2+) levels in ALL cells through constitutive Ca2+ entry and enhanced store-operated Ca2⁺ influx upon thapsigargin stimulation. This increase in basal Ca2+ signaling promoted ALL cell viability and proliferation in vitro. Notably, chelating intracellular Ca2+ with BAPTA-AM reduces GsMTx4-mediated leukemia cell viability and proliferation. However, in vivo, GsMTx4 decreases cytosolic Ca2+ levels in Nalm-6 GFP⁺ cells isolated from mouse blood, effectively countering leukemia progression and significantly extending survival in NSG mice transplanted with leukemia cells (median survival: GsMTx4 vs. control, 37.5 days vs. 29 days, p = 0.0414). Our results highlight the different properties of GsMTx4 activity in in vitro and in vivo models. They also emphasize that Ca2+ signaling is a key vulnerability in leukemia, where its precise modulation dictates disease progression. Thus, targeting Ca2+ channels could offer a novel therapeutic strategy for leukemia by exploiting Ca2+ homeostasis.
Keywords: ALL; Ca2+ signaling; GsMTx4; cell survival.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Improvement of dexamethasone sensitivity by chelation of intracellular Ca2+ in pediatric acute lymphoblastic leukemia cells through the prosurvival kinase ERK1/2 deactivation.Oncotarget. 2017 Apr 18;8(16):27339-27352. doi: 10.18632/oncotarget.16039. Oncotarget. 2017. PMID: 28423696 Free PMC article.
-
GsMTx4-blocked PIEZO1 channel promotes myogenic differentiation and alleviates myofiber damage in Duchenne muscular dystrophy.Skelet Muscle. 2025 May 14;15(1):13. doi: 10.1186/s13395-025-00383-5. Skelet Muscle. 2025. PMID: 40361216 Free PMC article.
-
Pyr3, a TRPC3 channel blocker, potentiates dexamethasone sensitivity and apoptosis in acute lymphoblastic leukemia cells by disturbing Ca(2+) signaling, mitochondrial membrane potential changes and reactive oxygen species production.Eur J Pharmacol. 2016 Aug 5;784:90-8. doi: 10.1016/j.ejphar.2016.05.014. Epub 2016 May 12. Eur J Pharmacol. 2016. PMID: 27179991
-
Pharmacological inhibition of store-operated calcium entry in MDA-MB-468 basal A breast cancer cells: consequences on calcium signalling, cell migration and proliferation.Cell Mol Life Sci. 2018 Dec;75(24):4525-4537. doi: 10.1007/s00018-018-2904-y. Epub 2018 Aug 13. Cell Mol Life Sci. 2018. PMID: 30105615 Free PMC article.
-
Mitochondria as emerging targets for therapies against T cell acute lymphoblastic leukemia.J Leukoc Biol. 2019 May;105(5):935-946. doi: 10.1002/JLB.5VMR0818-330RR. Epub 2019 Jan 30. J Leukoc Biol. 2019. PMID: 30698851 Review.
References
-
- Conter V., Aricò M., Basso G., Biondi A., Barisone E., Messina C., Parasole R., De Rossi G., Locatelli F., Pession A., et al. EDUCATIONAL REPORT Long-Term Results of the Italian Association of Pediatric Hematology and Oncology (AIEOP) Studies 82, 87, 88, 91 and 95 for Childhood Acute Lymphoblastic Leukemia. Leukemia. 2010;24:255–264. doi: 10.1038/leu.2009.250. - DOI - PubMed
-
- Serafin V., Capuzzo G., Milani G., Minuzzo S.A., Pinazza M., Bortolozzi R., Bresolin S., Porcù E., Frasson C., Indraccolo S., et al. Glucocorticoid Resistance Is Reverted by LCK Inhibition in Pediatric T-Cell Acute Lymphoblastic Leukemia. Blood. 2017;130:2750–2761. doi: 10.1182/blood-2017-05-784603. - DOI - PubMed
-
- Oskarsson T., Söderhäll S., Arvidson J., Forestier E., Frandsen T.L., Hellebostad M., Lähteenmäki P., Jónsson Ó.G., Myrberg I.H., Heyman M., et al. Treatment-Related Mortality in Relapsed Childhood Acute Lymphoblastic Leukemia. Pediatr. Blood Cancer. 2018;65:e26909. doi: 10.1002/pbc.26909. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

