Long-Term Supplementation of GABA Regulates Growth, Food Intake, Locomotion, and Lipid Metabolism by Increasing Ghrelin and Growth Hormone in Adolescent Mice
- PMID: 40431374
- PMCID: PMC12113763
- DOI: 10.3390/nu17101634
Long-Term Supplementation of GABA Regulates Growth, Food Intake, Locomotion, and Lipid Metabolism by Increasing Ghrelin and Growth Hormone in Adolescent Mice
Abstract
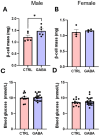
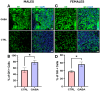
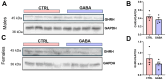
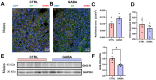
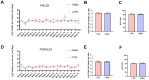
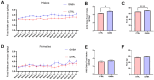
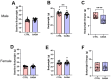
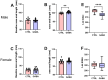
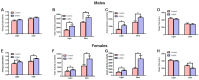
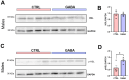
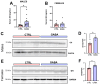
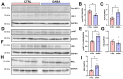
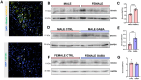
Background/Objectives: The amino acid γ-aminobutyric acid (GABA) is the primary neurotransmitter in the central nervous system (CNS) and acts as an autocrine and/or paracrine signaling molecule in various types of non-neuronal cells. On the other hand, GABA is a nutrient found in a variety of foods and is marketed as a health supplement based on a growing number of studies reporting health benefits in humans and recuperations in animal models of diseases. The present study sought to examine whether supplementation of GABA to young mice regulates their growth as well as glucose and lipid metabolism during physiological adolescence. Methods: Mice were supplemented with GABA over a 16-week period with subsequent anthropometric, metabolic, and endocrine measurements. Results: Results showed that 16-week oral supplementation of GABA increased food consumption and body length while attenuating weight gain in male mice but not females. In addition, GABA treatment lowered the index of body fat (Lee index) and increased the expression of lipolytic enzymes in adipose and liver tissues of male mice without affecting blood glucose levels. Remarkably, supplementation of GABA significantly increased the protein expression of growth hormone (GH) in the pituitary gland of both male and female mice. However, it only substantially increased GH levels in the sera of male mice but not females. Moreover, GABA significantly increased the expression of the GH secretagogue peptide ghrelin in the stomachs of male mice only. Conclusions: Together these novel findings suggest that long-term GABA supplementation fundamentally influences the growth and lipid metabolism of males during adolescent development by stimulating ghrelin-GH production and secretion. The mechanisms of GABA-induced sex-dependent upregulations of ghrelin and GH, as well as lipid metabolism in adolescence, await further studies.
Keywords: GABA; adolescent development; ghrelin; growth hormone; lipid metabolism.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Januzi L., Poirier J.W., Maksoud M.J.E., Xiang Y.-Y., Veldhuizen R.A.W., Gill S.E., Cregan S.P., Zhang H., Dekaban G.A., Lu W.-Y. Autocrine GABA Signaling Distinctively Regulates Phenotypic Activation of Mouse Pulmonary Macrophages. Cell. Immunol. 2018;332:7–23. doi: 10.1016/j.cellimm.2018.07.001. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

