Stromal Interferon Regulatory Factor 3 Can Antagonize Human Papillomavirus Replication by Supporting Epithelial-to-Mesenchymal Transition
- PMID: 40431610
- PMCID: PMC12115382
- DOI: 10.3390/v17050598
Stromal Interferon Regulatory Factor 3 Can Antagonize Human Papillomavirus Replication by Supporting Epithelial-to-Mesenchymal Transition
Abstract
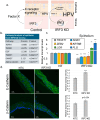
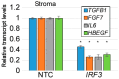
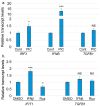
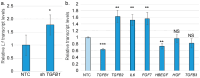
Epithelia contribute to the innate immune system through barrier formation and through signaling to immune cells. When the barrier is breached, epithelial cells undergo epithelial-to-mesenchymal transition (EMT) as part of the wound healing process. EMT is largely directed by signals from the stromal microenvironment, including transforming growth factor beta (TGFβ1), and antagonizes normal epithelial differentiation. How EMT and innate immunity may be connected molecularly has not been explored, although both processes are likely to occur simultaneously. Keratinocytes are the host cell type for human papillomaviruses (HPV), which can induce EMT in certain conditions but also depend on differentiation for their replication. We previously found that the innate immune factor interferon regulatory factor 3 (IRF3) inhibits epithelial differentiation and reduces the expression of HPV16 late genes. Here we report that IRF3 in the stroma compartment promotes an EMT-like pattern of gene expression in an HPV16-containing epithelium. The depletion of stromal IRF3 resulted in the downregulation of TGFβ1-related signaling in both the stroma and epithelium. IRF3 binds to the TGFB1 promoter in human foreskin fibroblasts and is necessary for TGFB1 mRNA production. Because an EMT-like state is unfavorable for differentiation-dependent HPV16, we observed that all EMT markers examined were reduced in the presence of episomal HPV16. Together, we show that stromal IRF3 can disrupt epithelial differentiation and act as an anti-HPV factor through the regulation of EMT, linking wound healing and immunity.
Keywords: epithelial-to-mesenchymal transition (EMT); human papillomavirus type 16 (HPV16); interferon regulatory factor 3 (IRF3); transforming growth factor beta 1 (TGFβ1).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Human papillomavirus 16 mitigates Sneathia vaginalis-induced damage to cervical keratinocytes.mSphere. 2025 Jul 29;10(7):e0015225. doi: 10.1128/msphere.00152-25. Epub 2025 Jul 1. mSphere. 2025. PMID: 40590527 Free PMC article.
-
The Human Papillomavirus E6 Oncoprotein Targets USP15 and TRIM25 To Suppress RIG-I-Mediated Innate Immune Signaling.J Virol. 2018 Feb 26;92(6):e01737-17. doi: 10.1128/JVI.01737-17. Print 2018 Mar 15. J Virol. 2018. PMID: 29263274 Free PMC article.
-
Microarray analysis of human keratinocytes from different anatomic sites reveals site-specific immune signaling and responses to human papillomavirus type 16 transfection.Mol Med. 2018 May 16;24(1):23. doi: 10.1186/s10020-018-0022-9. Mol Med. 2018. PMID: 30134802 Free PMC article.
-
Prophylactic vaccination against human papillomaviruses to prevent cervical cancer and its precursors.Cochrane Database Syst Rev. 2018 May 9;5(5):CD009069. doi: 10.1002/14651858.CD009069.pub3. Cochrane Database Syst Rev. 2018. PMID: 29740819 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
References
-
- Clark R.A. The Molecular and Cellular Biology of Wound Repair. Springer Science & Business Media; Berlin, Germany: 2013.
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

