Anxiety, depression, physical disease parameters and health-related quality of life in the BronchUK national bronchiectasis cohort
- PMID: 40432821
- PMCID: PMC12107378
- DOI: 10.1183/23120541.00348-2024
Anxiety, depression, physical disease parameters and health-related quality of life in the BronchUK national bronchiectasis cohort
Abstract
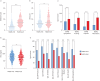
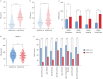
Background: Bronchiectasis is associated with psychological comorbidity and poor quality of life (QoL), yet guidelines lack focus on psychological morbidity. Using data obtained from the BronchUK database (1341 patients), we examined the link between anxiety/depression and physical disease severity, QoL and long-term outcomes in bronchiectasis.
Methods: Computed tomography-confirmed bronchiectasis patients enrolled in the BronchUK study with Hospital Anxiety and Depression Scale (HADS-A/D) data were studied. HADS-A/D scores ≥8 indicated anxiety/depression. QoL was measured by the St George's Respiratory Questionnaire and QoL-Bronchiectasis Questionnaire. Exacerbations during annual follow-up were analysed by negative binomial regression with time in study as an offset adjusted for age, body mass index, sex, Pseudomonas infection, diabetes and forced expiratory volume in 1 s (FEV1). Cox regression determined probability of hospitalisation using time to first exacerbation.
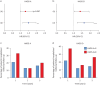
Results: 1341 patients were included; 418 had anxiety (31%), 269 (20%) had depression and 201 (15%) had both conditions. HADS-A/D ≥8 was associated with worse QoL (p<0.0001) and clinical severity (e.g. Bronchiectasis Severity Index, FEV1 and Medical Research Council dyspnoea score (all p<0.01). HADS-A/D ≥8 each was associated with exacerbation (rate ratio (RR) 1.42, 95% CI 1.32-1.52 for HADS-A; RR 1.45, 95% CI 1.34-1.56 for HADS-D, both p<0.0001) and hospitalisation risk (RR 1.58, 95% CI 1.29-1.92 for HADS-A; RR 1.76, 95% CI 1.43-2.17 for HADS-D, both p<0.001). HADS-A/D ≥8 each predicted future hospitalisation (HR 1.30, 95% CI 0.98-1.72, p=0.067 for HADS-A; HR 1.40 95% CI 1.04-1.88, p=0.027 for HADS-D).
Interpretation: Anxiety and depression are common in bronchiectasis, correlate with disease severity and predict poor outcomes. Consideration of psychological comorbidities should be evaluated in routine bronchiectasis care.
Copyright ©The authors 2025.
Conflict of interest statement
Conflict of interest: T. Saunders, J. Brown, A. Sullivan, M. Carroll, P. Mawson, T. Gatheral, A.T. Hill, G. Davies, J. Pollock, M. Kelly, R. McNally, G. Wild, V. Navarantnam and H. Upadhyay declare no conflict of interest in relation to this manuscript. Conflict of interest: A. De Soyza reports receiving funds from AstraZeneca, Byers, Forest, Gilead, Insmed, GSK and Novartis outside of work for this manuscript. Conflict of interest: S. Elborn reports research grants from iABC outside of work for this manuscript. Conflict of interest: C. Haworth reports consulting fees from 30 Technology, Aradigm, CSL Behring, Chiesi, Gilead, Grifols, GSK, Insmed, Janssen, LifeArc, Meiji, Mylan, Novartis, Pneumagen, Shionogi, Teva, Vertex and Zambon; payment from Chiesi, Grifols, GSK, Insmed, Mylan, Novartis, Teva, Vertex and Zambon; and support for attending meetings and/or travel from Zambon outside of work for this manuscript. Conflict of interest: J.R. Hurst reports consulting fees from AstraZeneca and GSK; payments from Boehringer Ingelheim, Chiesi, Sanofi and Takeda; support for attending meetings and travel from AstraZeneca; participation on an advisory board AstraZeneca; and receipt of equipment from Nonin outside of work for this manuscript. Conflict of interest: M. Loebinger reports consulting fees from Armata, 30T, AstraZeneca, Insmed, Cheisi, Zambon, Electromed, Recode, Boehringer Ingelheim, Ethris, Mannkind and AN2 Therapeutics; and received payment or honoraria for lectures or presentations from Insmed outside of work for this manuscript. Conflict of interest: J. Bradley reports research grants from Health and Social Care (Northern Ireland), iABC, NIHR and DfE; and receipt of equipment from PARI outside of work for this manuscript. Conflict of interest: P.P. Walker reports unpaid work as Chair of the Board, British Thoracic Society, outside of work for this manuscript. Conflict of interest: J. Steer reports research grants from Chiesi and Menarini Pharmaceutica; and speaker fees and conference and travel fees from AstraZeneca outside of work for this manuscript. Conflict of interest: J. Duckers reports consulting fees from Insmed; payment received from Vertex, Chiesi and Insmed; participation on a data safety monitoring board for NOMAB study; Chair of British Thoracic Society CF Specialist advisory Group, CF Trust Registry Steering group and South-East Wales Research Ethics Committee; and receipt of medical writing from IQVIA outside of work for this manuscript. Conflict of interest: M. Crichton declares consulting fees received from Boxer Capital LLC. Conflict of interest: J.D. Chalmers reports research grants from AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Gilead Sciences, Novartis and Insmed; and received consultancy or speaker fees from AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Insmed, Janssen, Novartis and Zambon outside of work for this manuscript; and is an associate editor of this journal.
Figures

Similar articles
-
Factors Associated With Quality of Life in Subjects With Stable COPD.Respir Care. 2015 Nov;60(11):1585-91. doi: 10.4187/respcare.03904. Epub 2015 Jul 7. Respir Care. 2015. PMID: 26152471
-
Anxiety and depression in adult outpatients with bronchiectasis: Associations with disease severity and health-related quality of life.Clin Respir J. 2018 Apr;12(4):1485-1494. doi: 10.1111/crj.12695. Epub 2017 Sep 19. Clin Respir J. 2018. PMID: 28842946
-
In bronchiectasis, poor physical capacity correlates with poor quality of life.Eur Clin Respir J. 2022 Jun 30;9(1):2095104. doi: 10.1080/20018525.2022.2095104. eCollection 2022. Eur Clin Respir J. 2022. PMID: 35800900 Free PMC article.
-
Exercise training for bronchiectasis.Cochrane Database Syst Rev. 2021 Apr 6;4(4):CD013110. doi: 10.1002/14651858.CD013110.pub2. Cochrane Database Syst Rev. 2021. PMID: 33822364 Free PMC article.
-
Adjuvant therapy with antidepressants for the management of inflammatory bowel disease.Cochrane Database Syst Rev. 2019 Apr 12;4(4):CD012680. doi: 10.1002/14651858.CD012680.pub2. Cochrane Database Syst Rev. 2019. PMID: 30977111 Free PMC article.
References
LinkOut - more resources
Full Text Sources
