Stromal vascular fraction inhibits renal fibrosis by regulating metabolism and inflammation in obstructive nephropathy
- PMID: 40432888
- PMCID: PMC12106432
- DOI: 10.3389/fphar.2025.1559446
Stromal vascular fraction inhibits renal fibrosis by regulating metabolism and inflammation in obstructive nephropathy
Abstract
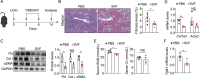
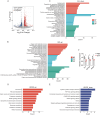
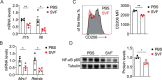
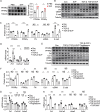
Obstructive nephropathy is one of the leading causes of kidney injury and fibrosis, which can lead to end-stage renal disease (ESRD). Stromal vascular fraction (SVF), a heterogeneous cell mixture derived from adipose tissue, has been widely used for regenerative medicine across many preclinical models and clinical applications. Recent studies have suggested that SVF can alleviate acute kidney injury in mice. However, to our knowledge, the therapeutic effects of SVF on obstructive nephropathy have not been studied before. In this study, we evaluated the therapeutic potential of SVF on obstructive nephropathy in mice with unilateral ureteral obstruction (UUO). We revealed that autologous SVF administration mitigated UUO-induced renal fibrosis. SVF treatment inhibited both the infiltration of neutrophils and CD4+ T cells, as well as the production of inflammatory cytokines. Moreover, SVF promoted metabolic reprogramming and improved mitochondrial function in the obstructed kidneys, partially through PPAR pathway activation. Mechanistically, SVF-mediated PPAR activation inhibited the epithelial-mesenchymal transition (EMT) process of tubular cells, thus alleviating renal fibrosis in UUO mice. We further confirmed that pharmacological activation of PPAR pathway significantly reduced fibrosis in UUO kidneys. Therefore, our study suggests that SVF may represent a promising therapeutic strategy for obstructive nephropathy.
Keywords: PPAR; SVF; inflammation; obstructive nephropathy; renal fibrosis.
Copyright © 2025 Yue, Yang, Jia, Wu, Ma, Yi, Tao and Zhou.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Dimethyl malonate alleviates obstructive nephropathy by enhancing renal metabolism and inhibiting kidney oxidative stress and inflammation.Front Pharmacol. 2025 Jun 10;16:1530635. doi: 10.3389/fphar.2025.1530635. eCollection 2025. Front Pharmacol. 2025. PMID: 40556756 Free PMC article.
-
Up-regulation of the human-specific CHRFAM7A gene protects against renal fibrosis in mice with obstructive nephropathy.J Cell Mol Med. 2023 Jan;27(1):52-65. doi: 10.1111/jcmm.17630. Epub 2022 Dec 7. J Cell Mol Med. 2023. PMID: 36479618 Free PMC article.
-
Preischemic Administration of Nonexpanded Adipose Stromal Vascular Fraction Attenuates Acute Renal Ischemia/Reperfusion Injury and Fibrosis.Stem Cells Transl Med. 2016 Sep;5(9):1277-88. doi: 10.5966/sctm.2015-0223. Epub 2016 Jun 30. Stem Cells Transl Med. 2016. PMID: 27365485 Free PMC article.
-
Unilateral Ureteral Obstruction as a Model to Investigate Fibrosis-Attenuating Treatments.Biomolecules. 2019 Apr 8;9(4):141. doi: 10.3390/biom9040141. Biomolecules. 2019. PMID: 30965656 Free PMC article. Review.
-
Stromal vascular fraction in canine osteoarthritis: advantages, applications, and insights for veterinary practitioners.Front Vet Sci. 2025 May 16;12:1586629. doi: 10.3389/fvets.2025.1586629. eCollection 2025. Front Vet Sci. 2025. PMID: 40454169 Free PMC article. Review.
References
-
- Bensemmane L., Milliat F., Treton X., Linard C. (2023). Systemically delivered adipose stromal vascular fraction mitigates radiation-induced gastrointestinal syndrome by immunomodulating the inflammatory response through a CD11b(+) cell-dependent mechanism. Stem Cell Res. Ther. 14 (1), 325. 10.1186/s13287-023-03562-7 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

