From Mechanoelectric Conversion to Tissue Regeneration: Translational Progress in Piezoelectric Materials
- PMID: 40434211
- PMCID: PMC12369703
- DOI: 10.1002/adma.202417564
From Mechanoelectric Conversion to Tissue Regeneration: Translational Progress in Piezoelectric Materials
Abstract
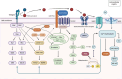
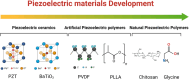
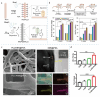
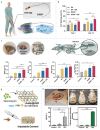
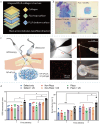
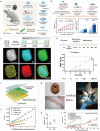
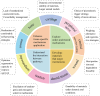
Piezoelectric materials, capable of converting mechanical stimuli into electrical signals, have emerged as promising tools in regenerative medicine due to their potential to stimulate tissue repair. Despite a surge in research on piezoelectric biomaterials, systematic insights to direct their translational optimization remain limited. This review addresses the current landscape by bridging fundamental principles with clinical potential. The biomimetic basis of piezoelectricity, key molecular pathways involved in the synergy between mechanical and electrical stimulation for enhanced tissue regeneration, and critical considerations for material optimization, structural design, and biosafety is discussed. More importantly, the current status and translational quagmire of mechanisms and applications in recent years are explored. A mechanism-driven strategy is proposed for the therapeutic application of piezoelectric biomaterials for tissue repair and identify future directions for accelerated clinical applications.
Keywords: advanced materials for translational medicine; biosensors; piezoelectricity; regenerative medicine; tissue engineering.
© 2025 The Author(s). Advanced Materials published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- F. D. I. S. C. O. B. Edition , FDA approval of Ryoncil (remestemcel‐L‐rknd) for steroid‐refractory acute graft versus host disease in pediatric patients, https://www.fda.gov/drugs/resources‐information‐approved‐drugs/fda‐disco... (accessed: February 2025).
-
- E. M. Agency , Alofisel withdrawn from the EU market, https://www.ema.europa.eu/en/news/alofisel‐withdrawn‐eu‐market (accessed: February 2025).
-
- B. Carmen Pope , Ryoncil, https://www.drugs.com/ryoncil.html (accessed: February 2025).
-
- a) Mao Y. L., Wickström S. A., Nat. Rev. Mol. Cell Biol. 2024; - PubMed
- b) Maurer M., Lammerding J., Annu. Rev. Biomed. Eng. 2019, 21, 443; - PMC - PubMed
- c) Di X., Gao X., Peng L., Ai J., Jin X., Qi S., Li H., Wang K., Luo D., Signal. Transduct. Target Ther. 2023, 8, 282; - PMC - PubMed
- d) Du H., Bartleson J. M., Butenko S., Alonso V., Liu W. F., Winer D. A., Butte M. J., Nat. Rev. Immunol. 2023, 23, 174. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

