Longitudinal Remote Sleep and Cognitive Research in Older Adults With Mild Cognitive Impairment and Dementia: Prospective Feasibility Cohort Study
- PMID: 40435500
- PMCID: PMC12159556
- DOI: 10.2196/72824
Longitudinal Remote Sleep and Cognitive Research in Older Adults With Mild Cognitive Impairment and Dementia: Prospective Feasibility Cohort Study
Abstract
Background: Sleep holds promise as a modifiable risk factor for neurodegenerative diseases and dementia. Clinical trials to modify sleep in people at risk of or in the early stages of dementia are needed. Monitoring natural sleep from home could support pragmatic and decentralized large-scale clinical trials. However, whether longitudinal sleep research can be successfully delivered remotely in this population has not been established yet.
Objective: We investigated the feasibility of remote longitudinal research using wearable devices, web-based cognitive tasks, and a smartphone app to record sleep and cognition in older adults with mild cognitive impairment (MCI) or dementia.
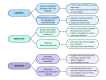
Methods: Older adults with MCI or dementia due to Alzheimer disease or Lewy body disease and cognitively healthy participants completed at-home sleep and circadian monitoring (digital sleep diaries, actigraphy, wearable sleep electroencephalography, and saliva samples) and digital cognitive assessments for 8 weeks. Feasibility outcomes included recruitment, retention, and data completeness.
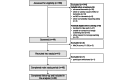
Results: In total, 41 participants consented (n=10, 24% participants with Alzheimer disease; n=11, 27% participants with Lewy body disease; and n=20, 49% controls). There were predominantly male and White British participants, with a mean age of 70.9 (SD 5.9) years. Retention was very high, with 40 (98%) participants completing 8 weeks of remote monitoring. Data completeness for sleep electroencephalography was 91% and ranged from 79% to 97% for all remote tasks and was overall high across all participant subgroups. In total, 30% (12/40) of participants reported receiving external support with completing study tasks.
Conclusions: High rates of retention, data completeness, and data quality suggested that longitudinal multimodal sleep and cognitive profiling using novel and remote monitoring technology is feasible in older adults with MCI and dementia and healthy older adults, even without study partner support. Remote monitoring should be considered for mechanistic and interventional trials. Careful consideration should be given to how to ensure remote monitoring technologies reduce burden and enhance inclusivity, particularly in communities traditionally underserved by research and those with lower digital literacy.
International registered report identifier (irrid): RR2-10.2196/52652.
Keywords: actigraphy; dementia; digital biomarkers; electroencephalography; feasibility; mild cognitive impairment; remote study design; saliva; sleep.
©Victoria Grace Gabb, Jonathan Blackman, Hamish Morrison, Haoxuan Li, Adrian Kendrick, Nicholas Turner, Rosemary Greenwood, Bijetri Biswas, Amanda Heslegrave, Elizabeth Coulthard. Originally published in JMIR Aging (https://aging.jmir.org), 28.05.2025.
Conflict of interest statement
Conflicts of Interest: EC has received funding from Biogen, Eisai, and Lilly for consultancy and providing educational resources. AH has received funding from Quanterix Corp for consultancy. All other authors declare no other conflicts of interest.
Figures


References
-
- Koren T, Fisher E, Webster L, Livingston G, Rapaport P. Prevalence of sleep disturbances in people with dementia living in the community: a systematic review and meta-analysis. Ageing Res Rev. 2023 Jan;83:101782. doi: 10.1016/j.arr.2022.101782. https://linkinghub.elsevier.com/retrieve/pii/S1568-1637(22)00224-0 S1568-1637(22)00224-0 - DOI - PubMed
-
- Postuma RB, Iranzo A, Hu M, Högl B, Boeve BF, Manni R, Oertel WH, Arnulf I, Ferini-Strambi L, Puligheddu M, Antelmi E, Cochen De Cock V, Arnaldi D, Mollenhauer B, Videnovic A, Sonka K, Jung KY, Kunz D, Dauvilliers Y, Provini F, Lewis SJ, Buskova J, Pavlova M, Heidbreder A, Montplaisir JY, Santamaria J, Barber TR, Stefani A, St Louis EK, Terzaghi M, Janzen A, Leu-Semenescu S, Plazzi G, Nobili F, Sixel-Doering F, Dusek P, Bes F, Cortelli P, Ehgoetz Martens K, Gagnon JF, Gaig C, Zucconi M, Trenkwalder C, Gan-Or Z, Lo C, Rolinski M, Mahlknecht P, Holzknecht E, Boeve AR, Teigen LN, Toscano G, Mayer G, Morbelli S, Dawson B, Pelletier A. Risk and predictors of dementia and parkinsonism in idiopathic REM sleep behaviour disorder: a multicentre study. Brain. 2019 Mar 01;142(3):744–59. doi: 10.1093/brain/awz030. https://europepmc.org/abstract/MED/30789229 5353011 - DOI - PMC - PubMed
-
- Harenbrock J, Holling H, Reid G, Koychev I. A meta-analysis of the relationship between sleep and β-Amyloid biomarkers in Alzheimer’s disease. Biomark Neuropsychiatry. 2023 Dec;9:100068. doi: 10.1016/j.bionps.2023.100068. - DOI
-
- Zamore Z, Veasey SC. Neural consequences of chronic sleep disruption. Trends Neurosci. 2022 Sep;45(9):678–91. doi: 10.1016/j.tins.2022.05.007. https://linkinghub.elsevier.com/retrieve/pii/S0166-2236(22)00101-1 S0166-2236(22)00101-1 - DOI - PMC - PubMed
-
- Zielinski MR, Gibbons AJ. Neuroinflammation, sleep, and circadian rhythms. Front Cell Infect Microbiol. 2022 Mar 22;12:853096. doi: 10.3389/fcimb.2022.853096. https://europepmc.org/abstract/MED/35392608 - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

