Repeated plasma p-tau217 measurements to monitor clinical progression heterogeneity
- PMID: 40442871
- PMCID: PMC12122256
- DOI: 10.1002/alz.70319
Repeated plasma p-tau217 measurements to monitor clinical progression heterogeneity
Abstract
Introduction: Heterogeneity of clinical progression in Alzheimer's disease (AD) complicates the assessment of disease progression and treatment effects in trials. This study evaluates the potential of plasma phosphorylated tau-217 (p-tau217) to capture this heterogeneity.
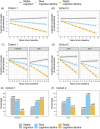
Methods: We used k-means clustering to analyze cognitive trajectories in amyloid beta -positive (Aβ+) cognitively normal (CN) and mild cognitive impairment (MCI) participants from two independent cohorts. Cohort 1 included 186 participants (71 CN, 115 MCI; 507 observations) and Cohort 2 included 207 participants (64 CN, 144 MCI; 781 observations), both with up to 10 years of follow-up.
Results: Three progression clusters emerged in both cohorts: stable cognition, slow decline, and rapid decline-each including cases initially classified as CN or MCI. Baseline plasma p-tau217 was linked to progression risk in both cohorts, whereas longitudinal increases in Cohort 1 were steepest in rapid decliners.
Discussion: Plasma p-tau217 may aid in capturing clinical heterogeneity and support stratification and monitoring of disease progression in clinical trials.
Highlights: k-Means found stable, slow, and rapid cognitive decline clusters in amyloid beta-positive (Aβ+) cases. Higher baseline plasma phosphorylated tau-217 (p-tau217) levels predicted faster cognitive decline. Longitudinal increases in plasma p-tau217 were steepest in rapid decliners. Plasma p-tau217 tracks clinical progression heterogeneity in Aβ+ cases. Cognitive stage and amyloid alone may miss severity and risk in early-stage Alzheimer's disease.
Keywords: Alzheimer's disease; amyloid; cerebrospinal fluid; clinical heterogeneity; clinical progression; clinical trials; plasma; p‐tau217.
© 2025 The Author(s). Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
B. E. K. has served as a consultant for Biogen and medical advisory boards for Biogen and Eli Lilly. H. Z. has served at scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZpath, Amylyx, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Enigma, LabCorp, Merry Life, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Quanterix, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave; has given lectures sponsored by Alzecure, BioArctic, Biogen, Cellectricon, Fujirebio, Lilly, Novo Nordisk, Roche, and WebMD; and is a co‐founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work). K.B. has served as a consultant and at advisory boards for Abbvie, AC Immune, ALZPath, AriBio, Beckman‐Coulter, BioArctic, Biogen, Eisai, Lilly, Moleac Pte. Ltd, Neurimmune, Novartis, Ono Pharma, Prothena, Quanterix, Roche Diagnostics, Sanofi and Siemens Healthineers; has served at data monitoring committees for Julius Clinical and Novartis; has given lectures, produced educational materials and participated in educational programs for AC Immune, Biogen, Celdara Medical, Eisai and Roche Diagnostics; and is a co‐founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program, outside the work presented in this article. T.F. has served as a consultant and on advisory boards for Biogen, Eisai, Novo Nordisk, Eli Lilly, and Roche. R.E.S. has served on advisory boards for Eisai and Eli Lilly. A.V.H. has participated in educational programs for Eisai and an educational advisory board for Lilly. The remainder of the authors report no relevant conflict of interest relevant for this publication. Author disclosures are available in the Supporting Information.
Figures

References
-
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer's disease. N Engl J Med. 2023;388:9‐21. - PubMed
MeSH terms
Substances
Grants and funding
- NRC 311993/Norwegian Research Council
- #2023-00356/Swedish Research Council
- #2022-01018/Swedish Research Council
- #2019-02397/Swedish Research Council
- 101053962/European Union's Horizon Europe research and innovation programme
- #ALFGBG-71320/Swedish State Support for Clinical Research
- #201809-2016862/Alzheimer Drug Discovery Foundation
- AD Strategic Fund
- #ADSF-21-831376-C/the Alzheimer's Association
- #ADSF-21-831381-C/the Alzheimer's Association
- #ADSF-21-831377-C/the Alzheimer's Association
- #ADSF-24-1284328-C/the Alzheimer's Association
- European Partnership on Metrology
- #22HLT07/Participating States
- Bluefield Project
- Cure Alzheimer's Fund
- Olav Thon Foundation
- Erling-Persson Family Foundation
- Stiftelsen för Gamla Tjänarinnor
- #FO2022-0270/Hjärnfonden
- 860197/Marie Skłodowska-Curie
- JPND2021-00694/European Union Joint Programme - Neurodegenerative Disease Research
- National Institute for Health
- Care Research University College London Hospitals Biomedical Research Centre
- UKDRI-1003/UK Dementia Research Institute
- #2017-00915/UK Dementia Research Institute
- #2022-00732/UK Dementia Research Institute
- #AF-930351/Swedish Alzheimer Foundation
- #AF-939721/Swedish Alzheimer Foundation
- #AF-968270/Swedish Alzheimer Foundation
- #AF-994551/Swedish Alzheimer Foundation
- #ALZ2022-0006/Swedish Alzheimer Foundation
- #FO2024-0048-TK-130/Swedish Alzheimer Foundation
- #ALFGBG-965240/Swedish Alzheimer Foundation
- #ALFGBG-1006418/Swedish Alzheimer Foundation
- JPND2019-466-236/European Union Joint Program for Neurodegenerative Disorders
- ZEN-21-848495/European Union Joint Program for Neurodegenerative Disorders
- SG-23-1038904 QC/European Union Joint Program for Neurodegenerative Disorders
- La Fondation Recherche Alzheimer (FRA)
- Kirsten and Freddy Johansen Foundation
- Copenhagen
- Familjen Rönströms Stiftelse
LinkOut - more resources
Full Text Sources
Medical

