Galectin-9 induces IL-1β production as a key inflammatory cytokine in the acute myeloid leukemia cell line (U937)
- PMID: 40444162
- PMCID: PMC12118778
- DOI: 10.4103/RPS.RPS_234_23
Galectin-9 induces IL-1β production as a key inflammatory cytokine in the acute myeloid leukemia cell line (U937)
Abstract
Background and purpose: T-cell immunoglobulin and mucin-domain containing protein-3 (TIM-3)/ galectin-9 (Gal-9)/ autocrine loop in myeloid leukemia stem cells provokes inflammation through the NF-κB signaling pathway, which is influential in the expression of inflammatory factors. Interleukin 1β (IL-1β) is a vital inflammatory cytokine that plays an important role in the proliferation and therapy resistance of acute myeloid leukemia (AML) cells. This study aimed to assess the effect of Gal-9 on IL-1β in the human leukemic U937 cell line.
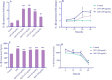
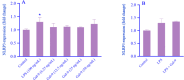
Experimental approach: The U937 cells were cultured in different concentrations of Gal-9. Cell counting kit-8 was used to assess the effect of Gal-9 on human leukemic U937 cell proliferation. Also, its impact on the expression of TIM-3, Gal-9, IL-1β, IL-1βR, IL-1βRAP, and NLRP3 genes and IL-1β protein was studied by RT-PCR and ELISA, respectively. Moreover, the effect of Gal-9 on the NF-κB signaling pathway was evaluated by western blotting.
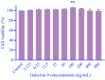
Findings/results: U937 cells were expanded in the presence of Gal-9 in a concentration-dependent manner. Following treatment of U937 cells with Gal-9, the gene expression of Gal-9, IL-1B, IL-1BR, and IL-1BRAP were significantly upregulated compared to the control group. The IL-1β concentration increased following Gal-9 treatment in a concentration-dependent manner, while following time-pass its level significantly decreased. Furthermore, Gal-9 slightly increased NF-κB phosphorylation.
Conclusion and implications: Gal-9 increased IL-1β level as a critical inflammatory cytokine in the proliferation and resistance of AML cells to therapy. According to this finding, targeting and blocking the TIM-3/Gal-9 autocrine loop can suppress IL-1β production and facilitate AML treatment.
Keywords: AML; GAL-9; IL-1β; Inflammation; NF-κB.
Copyright: © 2025 Research in Pharmaceutical Sciences.
Conflict of interest statement
The authors declared no conflict of interest in this study.
Figures




Similar articles
-
Oridonin Could Inhibit Inflammation and T-cell Immunoglobulin and Mucin-3/Galectin-9 (TIM-3/Gal-9) Autocrine Loop in the Acute Myeloid Leukemia Cell Line (U937) as Compared to Doxorubicin.Iran J Allergy Asthma Immunol. 2020 Dec 19;19(6):602-611. doi: 10.18502/ijaai.v19i6.4929. Iran J Allergy Asthma Immunol. 2020. PMID: 33463129
-
A TIM-3/Gal-9 Autocrine Stimulatory Loop Drives Self-Renewal of Human Myeloid Leukemia Stem Cells and Leukemic Progression.Cell Stem Cell. 2015 Sep 3;17(3):341-52. doi: 10.1016/j.stem.2015.07.011. Epub 2015 Aug 13. Cell Stem Cell. 2015. PMID: 26279267
-
[A TIM-3/galectin-9 autocrine stimulatory loop drives self-renewal of human myeloid leukemia stem cells and leukemia progression].Rinsho Ketsueki. 2016 Apr;57(4):412-6. doi: 10.11406/rinketsu.57.412. Rinsho Ketsueki. 2016. PMID: 27169443 Japanese.
-
Effect of exogenous galectin-9, a natural TIM-3 ligand, on the severity of TNBS- and DSS-induced colitis in mice.Int Immunopharmacol. 2023 Feb;115:109645. doi: 10.1016/j.intimp.2022.109645. Epub 2023 Jan 5. Int Immunopharmacol. 2023. PMID: 36610329
-
Phenylarsine oxide blocks interleukin-1beta-induced activation of the nuclear transcription factor NF-kappaB, inhibits proliferation, and induces apoptosis of acute myelogenous leukemia cells.Blood. 1999 Oct 15;94(8):2844-53. Blood. 1999. PMID: 10515888
References
-
- Winer ES. Secondary acute myeloid leukemia: a primary challenge of diagnosis and treatment. Hematol Oncol Clin North Am. 2020;34(2):449–463. DOI: 10.1016/j.hoc.2019.11.003. - PubMed
-
- Strickland SA, Vey N. Diagnosis and treatment of therapy-related acute myeloid leukemia. Crit Rev Oncol Hematol. 2022;171:103607. 1-10. DOI: 10.1016/j.critrevonc.2022.103607. - PubMed
-
- Bullinger L, Dohner K, Dohner H. Genomics of acute myeloid leukemia diagnosis and pathways. J Clin Oncol. 2017;35(9):934–946. DOI: 10.1200/JCO.2016.71.2208. - PubMed
-
- DiNardo CD, Wei AH. How I treat acute myeloid leukemia in the era of new drugs. Blood. 2020;135(2):85–96. DOI: 10.1182/blood.2019001239. - PubMed
-
- Short NJ, Konopleva M, Kadia TM, Borthakur G, Ravandi F, DiNardo CD, et al. Advances in the treatment of acute myeloid leukemia: new drugs and new challenges. Cancer Discov. 2020;10(4):506–525. DOI: 10.1158/2159-8290.CD-19-1011. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
