Efficient cell-free translation from diverse human cell types
- PMID: 40447189
- PMCID: PMC12226075
- DOI: 10.1016/j.jbc.2025.110307
Efficient cell-free translation from diverse human cell types
Abstract
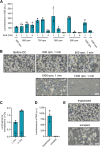
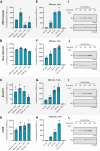
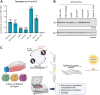
Cell-free translation systems are indispensable for studying protein synthesis, enabling researchers to explore translational regulation across different cell types. The difficulties in producing cell-free translation systems from different cell types limit the ability to study regulatory mechanisms that depend on different biological contexts. Here, we present a scalable method for preparing translation-competent lysates from a range of frequently used human cell lines using dual centrifugation. We optimized lysis conditions for adherent and suspension cells, producing high-quality lysates from HEK-293 (adherent and in suspension), HeLa, SH-SY5Y, and U2OS cells. Our results demonstrate that cell-specific factors influence translation efficiency, with adherent HeLa cells showing the highest activity. We also observed that sensitivity to lysis conditions varies between cell lines, underscoring the importance of fine-tuning parameters for efficient protein production. Our method provides a robust and adaptable approach for generating cell-type-specific lysates, broadening the application of in vitro translation systems in studying translational mechanisms.
Keywords: cell-free translation; cell-type-specific lysates; dual centrifugation; in vitro translation; protein synthesis; synthetic biology; translational regulation.
Copyright © 2025 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article.
Figures

Similar articles
-
Factors that influence the provision of intrapartum and postnatal care by skilled birth attendants in low- and middle-income countries: a qualitative evidence synthesis.Cochrane Database Syst Rev. 2017 Nov 17;11(11):CD011558. doi: 10.1002/14651858.CD011558.pub2. Cochrane Database Syst Rev. 2017. PMID: 29148566 Free PMC article.
-
Factors that influence caregivers' and adolescents' views and practices regarding human papillomavirus (HPV) vaccination for adolescents: a qualitative evidence synthesis.Cochrane Database Syst Rev. 2025 Apr 15;4(4):CD013430. doi: 10.1002/14651858.CD013430.pub2. Cochrane Database Syst Rev. 2025. PMID: 40232221 Free PMC article.
-
Falls prevention interventions for community-dwelling older adults: systematic review and meta-analysis of benefits, harms, and patient values and preferences.Syst Rev. 2024 Nov 26;13(1):289. doi: 10.1186/s13643-024-02681-3. Syst Rev. 2024. PMID: 39593159 Free PMC article.
-
Leveraging a foundation model zoo for cell similarity search in oncological microscopy across devices.Front Oncol. 2025 Jun 18;15:1480384. doi: 10.3389/fonc.2025.1480384. eCollection 2025. Front Oncol. 2025. PMID: 40606969 Free PMC article.
-
Factors that influence participation in physical activity for people with bipolar disorder: a synthesis of qualitative evidence.Cochrane Database Syst Rev. 2024 Jun 4;6(6):CD013557. doi: 10.1002/14651858.CD013557.pub2. Cochrane Database Syst Rev. 2024. PMID: 38837220 Free PMC article. Review.
References
-
- Swartz J. Developing cell-free biology for industrial applications. J. Ind. Microbiol. Biotechnol. 2006;33:476–485. - PubMed
-
- Gao M., Wang D., Wilsch-Bräuninger M., Leng W., Schulte J., Morgner N., et al. Cell free expression in proteinosomes prepared from native protein-PNIPAAm conjugates. Macromolecular Biosci. 2024;24 - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

