Patellofemoral Joint Chondral Defects Treated With Third-Generation Matrix-Induced Autologous Chondrocyte Implantation on Porcine Collagen Membrane: Minimum 2-Year Follow-up
- PMID: 40453957
- PMCID: PMC12123140
- DOI: 10.1177/23259671251341474
Patellofemoral Joint Chondral Defects Treated With Third-Generation Matrix-Induced Autologous Chondrocyte Implantation on Porcine Collagen Membrane: Minimum 2-Year Follow-up
Abstract
Background: Third-generation autologous chondrocyte implantation (ACI), also known as matrix-induced ACI (MACI), was approved for clinical practice in December 2016. Studies specifically investigating outcomes of MACI for complex patellofemoral chondral lesions are limited.
Purpose: To report patient-reported outcome measures (PROMs), complications, and failure rates after MACI for patellofemoral chondral defects at a minimum follow-up of 2 years.
Study design: Case series; Level of evidence, 4.
Methods: For this retrospective review of prospectively collected data, the authors identified patients who underwent treatment with MACI for focal chondral defects in the knee between August 2017 and September 2020. PROMs, including International Knee Documentation Committee (IKDC) score, Kujala score for patellofemoral disorders, and the Veterans RAND 12-item Health Survey (VR-12) score, were obtained preoperatively and a minimum of 2 years postoperatively. The percentage of patients who met the minimal clinically important difference (MCID) for each PROM was reported. Failure was defined as (1) graft failure on follow-up magnetic resonance imaging or second-look arthroscopy, (2) revision MACI or other chondral procedure, or (3) conversion to unicompartmental or total knee arthroplasty.
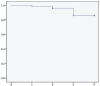
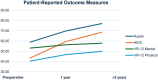
Results: A total of 50 patients (34 female; mean age, 32.43 ± 7.33 years; mean follow-up, 2.71 ± 0.79 years) remained after application of the exclusion criteria. There was a significant increase in all PROMs from preoperatively to postoperatively, including the IKDC (43.39 vs 68.58; P < .001), Kujala (58.93 vs 77.07; P < .001), VR-12 Mental (53.12 vs 57.90; P = .002), and VR-12 Physical (40.40 vs 49.89; P < .001) scores, with 5 (10%) failures. The MCID was achieved by 77.8% of patients in IKDC score and 69.0% in the Kujala score. Kaplan-Meier survival analysis showed survival probabilities of 98.0%, 96.0%, and 85.7% at 1, 2, and 4 years, respectively. MACI for patellofemoral bipolar lesions (n = 11) showed significant improvement in IKDC (50.06 vs 74.07; P = .008) and Kujala (69.33 vs 84.33; P = .046) scores, and 2 (18.2%) failures. Kaplan-Meier survival analysis with log-rank test demonstrated no significant differences in survival distributions between unipolar and bipolar patellofemoral lesions (P = .387).
Conclusions: Third-generation ACI (MACI) is a successful and effective treatment method for difficult-to-treat patellar, trochlear, and bipolar patellofemoral chondral defects.
Keywords: articular cartilage; articular cartilage resurfacing; autologous chondrocyte implantation; chondral defect; knee; patella.
© The Author(s) 2025.
Conflict of interest statement
One or more of the authors has declared the following potential conflict of interest or source of funding: A.H.G. has received royalties from Moximed Inc and Organogenesis; consulting fees from Bioventus LLC, Flexion Therapeutics Inc, Joint Restoration Foundation Inc, Miach Orthopaedics Inc, Moximed Inc, Organogenesis Inc, Smith & Nephew Inc, and Vericel Corporation; hospitality payments from Aesculap Biologics LLC, Bioventus LLC, Cartiheal Inc, DePuy Synthes Sales Inc, Flexion Therapeutics Inc, Joint Restoration Foundation Inc, Linvatec Corporation, Miach Orthopaedics Inc, Organogenesis, Pacira Therapeutics, and Smith & Nephew Inc; honoraria from Fidia Pharma USA Inc, Joint Restoration Foundation Inc, and Vericel Corporation; acquisitions from Smith & Nephew Inc; research support from Aesculap Biologics LLC, Cartiheal Inc, Hyalex Orthopaedics Inc, Joint Restoration Foundation Inc, Miach Orthopaedics Inc, Moximed Inc, Organogenesis Inc, Smith & Nephew Inc, and Vericel Corporation; and is a paid presenter or speaker for Linvatec Corporation, Pacira Therapeutics, Smith & Nephew Inc, and Vericel Corporation; he holds stock or stock options in Engage Uni LLC and Smith & Nephew Inc. S.M.S. has received hospitality payments from Bioventus LLC, Cartiheal Inc, DePuy Synthes Sales Inc, Dynasplint Systems Inc, Flexion Therapeutics Inc, Joint Restoration Foundation Inc, Linvatec Corporation, Miach Orthopaedics Inc, Organogenesis Inc, Pacira Therapeutics, Smith & Nephew Inc, and Vericel Corporation; research support from Cartiheal Inc, Hyalex Orthopaedics Inc, Joint Restoration Foundation Inc, Miach Orthopaedics Inc, Moximed Inc, Organogenesis Inc, Smith & Nephew Inc, and Vericel Corporation; honoraria from Joint Restoration Foundation Inc and Vericel Corporation; consulting fees from Bioventus LLC, Flexion Therapeutics Inc, Miach Orthopaedics Inc, Moximed Inc, Smith & Nephew Inc, and Vericel Corporation; and is a paid presenter or speaker for Smith & Nephew Inc and Vericel; she holds stock or stock options in Engage Uni LLC, Moximed Inc, and Stryker. A.H.G. and S.M.S. are spouses. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto. Ethical approval for this study was obtained from the Hospital for Special Surgery Institutional Review Board (2020-2123-AM35).
Figures

Similar articles
-
Management of patellar and trochlear cartilage lesions with matrix-induced autologous chondrocyte implantation in conjunction with patellofemoral realignment procedures improves patient-reported outcomes and magnetic resonance image appearance.J ISAKOS. 2024 Oct;9(5):100311. doi: 10.1016/j.jisako.2024.100311. Epub 2024 Aug 21. J ISAKOS. 2024. PMID: 39154863
-
Clinical and Radiological Outcomes at ≥10-Year Follow-up After Matrix-induced Autologous Chondrocyte Implantation in the Patellofemoral Joint.Am J Sports Med. 2024 Aug;52(10):2532-2540. doi: 10.1177/03635465241262337. Epub 2024 Aug 5. Am J Sports Med. 2024. PMID: 39101611 Free PMC article.
-
Impact of Wiberg Patellar Type on Outcomes and Survival Following Cell-Based Cartilage Repair for Patellar Chondral Lesions at Midterm Follow-up.Am J Sports Med. 2024 Mar;52(3):705-709. doi: 10.1177/03635465231220028. Epub 2024 Jan 20. Am J Sports Med. 2024. PMID: 38243799 Free PMC article.
-
Minimum 10-Year Outcomes of Matrix-Induced Autologous Chondrocyte Implantation in the Knee: A Systematic Review.Am J Sports Med. 2024 Jul;52(9):2407-2414. doi: 10.1177/03635465231205309. Epub 2024 Feb 5. Am J Sports Med. 2024. PMID: 38312085
-
Comparative Effectiveness of Cartilage Repair With Respect to the Minimal Clinically Important Difference.Am J Sports Med. 2019 Nov;47(13):3284-3293. doi: 10.1177/0363546518824552. Epub 2019 May 13. Am J Sports Med. 2019. PMID: 31082325
References
-
- Barié A, Kruck P, Sorbi R, et al.. Prospective long-term follow-up of autologous chondrocyte implantation with periosteum versus matrix-associated autologous chondrocyte implantation: a randomized clinical trial. Am J Sports Med. 2020;48(9):2230-2241. - PubMed
-
- Bedi A, Feeley BT, Williams RJ. Management of articular cartilage defects of the knee. J Bone Joint Surg Am. 2010;92(4):994-1009. - PubMed
-
- Bhosale AM, Richardson JB. Articular cartilage: structure, injuries and review of management. Br Med Bull. 2008;87:77-95. - PubMed
-
- Brittberg M. Cell carriers as the next generation of cell therapy for cartilage repair: a review of the matrix-induced autologous chondrocyte implantation procedure. Am J Sports Med. 2010;38(6):1259-1271. - PubMed
-
- Brittberg M, Recker D, Ilgenfritz J, Saris DBF; SUMMIT Extension Study Group. Matrix-applied characterized autologous cultured chondrocytes versus microfracture: five-year follow-up of a prospective randomized trial. Am J Sports Med. 2018;46(6):1343-1351. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

