Venetoclax and hypomethylating agents in octogenarians and nonagenarians with acute myeloid leukemia
- PMID: 40454395
- PMCID: PMC12082125
- DOI: 10.1016/j.bneo.2024.100016
Venetoclax and hypomethylating agents in octogenarians and nonagenarians with acute myeloid leukemia
Abstract
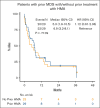
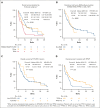
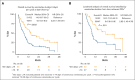
Venetoclax (VEN) plus a hypomethylating agent (HMA) regimen is the standard of care for older adults with acute myeloid leukemia (AML); however, it is associated with significant myelosuppression and complications, potentially limiting its use in those who are very old. We performed a multicenter retrospective analysis of VEN-HMA treatment in octogenarians and nonagenarians to further understand the tolerability, feasibility, dosing considerations, and clinical efficacy in this unique group. Patients with AML aged ≥80 years who received VEN-HMA between March 2015 and April 2022 were reviewed. VEN-HMA dosing was determined by treating physician, accounting for CYP3A4 drug interaction dose adjustments. In total, 154 patients were included, with a median age of 82 years (range, 80-92), who received treatment with VEN-HMA (83% with azacitidine and 17% with decitabine). Most patients (53%) had European LeukemiaNet 2017 adverse risk AML, 33% had intermediate, 8% had favorable, and 6% were unknown. With a median follow-up of 7.7 months, 36 patients (23%) remained in remission, with 31 (20%) still on VEN-HMA. The 30-day and 60-day mortality rates were 8.5% and 17%, respectively. The composite complete remission (CRc) rate for patients with newly diagnosed AML without prior myelodysplastic syndrome was 73% (48 of 66). Median overall survival (OS) was 8.1 months, and in patients who achieved a response (CRc), median OS was 13.2 months. Landmark analysis from the time CRc was first achieved showed that patients receiving VEN for ≤14 days had improved OS; median, 24.0 months. Patients who are very old can be treated safely with combination VEN-HMA with expectations of dose reductions and cycle extensions to ensure tolerability over the long term.
© 2024 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
Conflict-of-interest disclosure: M.A.S. reports role on advisory boards of Bristol Myers Squibb, Kurome, and Novartis. T.B. reports role on advisory boards of MorphoSys, Sobi, Servier, and Novartis; and reports consulting role with Novartis. N.S.C. reports salary support funded by Gilead Biosciences as a part of Winn Diversity in Clinical Trials Award 2023-2025. S.G.I. served on the advisory board of MorphoSys. A.K. reports consultancy with Geron, Sobi, Servier, MorphoSys, and GM Pharma; served as consultant/speaker bureau member of AbbVie, Astellas, AstraZeneca, Immunogen, Janssen, Menarini/Stemline, Pfizer, Ryvu, Servier, Syros, and Takeda; and reports research support from AbbVie, Astellas, AstraZeneca, Daiichi Sankyo, Pfizer, and Syros. D.A.S. reports consultancy with AbbVie, Affimed Gmbh, Gilead, Incyte, Intellisphere LLC, Molecular Partners AG, PGEN Therapeutics, Inc, Takeda, and Zentalis; served on the advisory board of AvenCell, bluebird bio, Bristol Myers Squibb, Intellia, Jasper Therapeutics, Kite, Magenta Therapeutics, NKARTA, Novartis, Orbital Therapeutics, Shattuck Labs, Servier, Syndax, and Syros; and reports research funding from Aprea and Jazz. D.A.P. served as an advisory board member of, and/or consultant for, Aptevo, Rigel, Novartis, Sumitomo, Adicet, AbbVie, Syros, Qihan, Seres, Gilead, OncoVerity, Bristol Myers Squibb, Boheringer Ingelheim, Sanofi, Karyopharm, MEI, Rigel, Syndax, and Treadwell; and reports research funding from AbbVie, Teva, Karyopharm, and Bristol Myers Squibb. M.R.S. received research funding from ALX Oncology, Astex, Incyte, Takeda, and TG Therapeutics; has equity in Empath Bio, Karyopharm, and Ryvu; reports advisory board/consulting role for AbbVie, Bristol Myers Squibb, Forma, Geron, GlaxoSmithKline, Karyopharm, Ryvu, and Taiho. C.P. reports advisory board role with Pfizer, Astellas, Janssen, GlaxoSmithKline, Blueprint, Jazz Pharmaceuticals, AbbVie, Novartis, and Delbert Laboratoires; and received honoraria from AbbVie, Astellas, Servier, Menarini/Stemline, Bristol Myers Squibb, Pfizer, Amgen, Janssen, Incyte, and Novartis. R.K. reports advisory board role with Taiho, Rigel, and DS InPharmatics; reports speaker’s bureau/advisory board role with AbbVie, CTI biopharma, Jazz, Pharma Essentia, and Servio; served as consultant for Geron; and received a research grant from Bristol Myers Squibb. J.W. reports advisory board/consultancy role with Servier, Rigel, Bristol Myers Squibb, Aptose, Daiichi Sankyo, and Ativarre; and reports research support from Takeda, Rigel, and Immune Systems Key Ltd. The remaining authors declare no competing financial interests.
Figures
References
LinkOut - more resources
Full Text Sources
Miscellaneous