Cerebrospinal fluid attenuates the efficacy of methotrexate against acute lymphoblastic leukemia cells
- PMID: 40454410
- PMCID: PMC12082133
- DOI: 10.1016/j.bneo.2024.100057
Cerebrospinal fluid attenuates the efficacy of methotrexate against acute lymphoblastic leukemia cells
Abstract
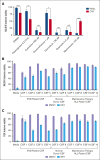
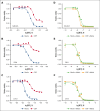
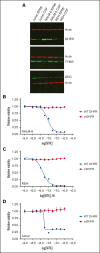
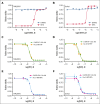
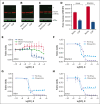
Treatment and prophylaxis of the central nervous system (CNS) is a standard component of acute lymphoblastic leukemia (ALL) therapy. However, CNS-directed therapies are a significant cause of morbidity, and CNS relapse remains a cause of treatment failure. CNS-directed ALL therapies must target leukemia cells within cerebrospinal fluid (CSF), a fluid that is compositionally distinct from plasma and has been shown to affect leukemia biology. Herein, we demonstrate that human CSF attenuates the potency and efficacy of antifolate drugs including methotrexate, the primary CNS-directed chemotherapeutic for >6 decades. Importantly, this effect of CSF on leukemia methotrexate sensitivity was reversible. Additional mechanistic studies support that diminished proliferation and activation of the integrated stress response in leukemia cells in the CSF may contribute to this resistance. Our findings suggest potential strategies to enhance methotrexate efficacy in CNS-directed ALL therapy and highlight the need to critically reassess even established standards of care.
© 2024 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures


Similar articles
-
Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis.Cochrane Database Syst Rev. 2020 Oct 19;10(10):CD012859. doi: 10.1002/14651858.CD012859.pub2. Cochrane Database Syst Rev. 2020. PMID: 33075160 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Restrictive versus liberal red blood cell transfusion strategies for people with haematological malignancies treated with intensive chemotherapy or radiotherapy, or both, with or without haematopoietic stem cell support.Cochrane Database Syst Rev. 2017 Jan 27;1(1):CD011305. doi: 10.1002/14651858.CD011305.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2024 May 23;5:CD011305. doi: 10.1002/14651858.CD011305.pub3. PMID: 28128441 Free PMC article. Updated.
-
Antidepressants for pain management in adults with chronic pain: a network meta-analysis.Health Technol Assess. 2024 Oct;28(62):1-155. doi: 10.3310/MKRT2948. Health Technol Assess. 2024. PMID: 39367772 Free PMC article.
-
Acute leukemia in children: evaluating the necessity of routine biochemical analysis of cerebrospinal fluid.Eur J Pediatr. 2025 Jan 20;184(2):148. doi: 10.1007/s00431-025-05986-0. Eur J Pediatr. 2025. PMID: 39831908
References
-
- Whiteside JA, Philips FS, Dargeon HW, Burchenal JH. Intrathecal amethopterin in neurological manifestations of leukemia. AMA Arch Intern Med. 1958;101(2):279–285. - PubMed
-
- Balis FM, Poplack DG. Central nervous system pharmacology of antileukemic drugs. Am J Pediatr Hematol Oncol. 1989;11(1):74–86. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

