Tumor-intrinsic TTLL12 drives resistance to cancer immunotherapy via modulating myeloid-derived suppressor cells
- PMID: 40461158
- PMCID: PMC12142039
- DOI: 10.1136/jitc-2024-010873
Tumor-intrinsic TTLL12 drives resistance to cancer immunotherapy via modulating myeloid-derived suppressor cells
Abstract
Background: The majority of patients treated with immunotherapy as a standalone therapy experience little to no benefits. Tubulin tyrosine ligase 12 (TTLL12), as a member of the tubulin tyrosine ligase protein family, is associated with the prognosis of patients with cancer and implicated in regulating innate immunity. However, the role of TTLL12 in modulating antitumor immunity remains unclear.
Methods: We analyzed the expression of TTLL12 in several cancer types and evaluated the putative correlation between TTLL12 expression and the immune infiltration in our own proteomic profile of human colorectal cancer. The gain-of-function or loss-of-function approaches were then implemented in vitro and in vivo, followed by flow cytometry to quantify immune cell population frequency. In vitro assays were used to confirm the influence of TTLL12 on the migration and proliferation of the immune subset. Mass spectrometry and chromatin-immunoprecipitation were used to further explore how TTLL12 influenced the immune subset. Finally, whether TTLL12 could enhance the antitumor efficacy of anti-programmed cell death protein 1 (PD-1) therapy was assessed in an immunocompetent mouse model.
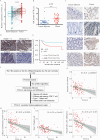
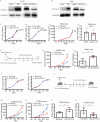
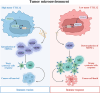
Results: We demonstrated that TTLL12 was upregulated in several cancer types and was linked with poor prognosis in patients with colorectal cancer. TTLL12 was found to be negatively correlated with the immune effector signature on the protein level in our proteomic datasets. TTLL12 ectopic expression in tumor cells did not influence cell proliferation but promoted tumor progression in syngeneic mouse models by modulating myeloid-derived suppressor cells (MDSCs), resulting in a suppressive immune response. TTLL12 was further proved to enrich MDSCs by promoting MDSC migration and proliferation in vitro. Mechanistically, tumor-derived TTLL12 induced the secretion of chemokine CCL9 via promoting its transcription, probably through binding to the promoter region of CCL9. Downregulating TTLL12 could significantly improve the efficacy of anti-PD-1 therapy in an immunocompetent murine model.
Conclusion: We identify TTLL12 as a key determinant of suppressive immune response, highlighting a previously unknown therapeutic target to increase the efficacy of antitumor immunotherapy.
Keywords: Cytokine; Immunotherapy; Myeloid-derived suppressor cell - MDSC.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ Group.
Conflict of interest statement
Competing interests: None declared.
Figures






References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
