Cysteine depletion triggers adipose tissue thermogenesis and weight loss
- PMID: 40461845
- PMCID: PMC12198010
- DOI: 10.1038/s42255-025-01297-8
Cysteine depletion triggers adipose tissue thermogenesis and weight loss
Abstract
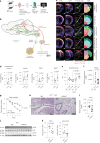
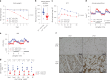
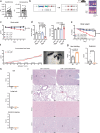
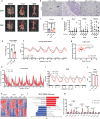
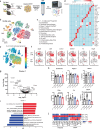
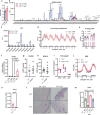
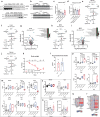
Caloric restriction and methionine restriction-driven enhanced lifespan and healthspan induces 'browning' of white adipose tissue, a metabolic response that increases heat production to defend core body temperature. However, how specific dietary amino acids control adipose thermogenesis is unknown. Here, we identified that weight loss induced by caloric restriction in humans reduces thiol-containing sulfur amino acid cysteine in white adipose tissue. Systemic cysteine depletion in mice causes lethal weight loss with increased fat utilization and browning of adipocytes that is rescued upon restoration of cysteine in diet. Mechanistically, cysteine-restriction-induced adipose browning and weight loss requires sympathetic nervous system-derived noradrenaline signalling via β3-adrenergic-receptors that is independent of FGF21 and UCP1. In obese mice, cysteine deprivation induced rapid adipose browning, increased energy expenditure leading to 30% weight loss and reversed metabolic inflammation. These findings establish that cysteine is essential for organismal metabolism as removal of cysteine in the host triggers adipose browning and rapid weight loss.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures






Update of
-
[WITHDRAWN] Activation of transsulfuration pathway to maintain cysteine is a thermogenic checkpoint for the conservation of energy.Res Sq [Preprint]. 2023 Jun 30:rs.3.rs-3069713. doi: 10.21203/rs.3.rs-3069713/v1. Res Sq. 2023. Update in: Nat Metab. 2025 Jun;7(6):1204-1222. doi: 10.1038/s42255-025-01297-8. PMID: 37461682 Free PMC article. Updated. Preprint.
-
Cysteine depletion triggers adipose tissue thermogenesis and weight-loss.bioRxiv [Preprint]. 2024 Aug 8:2024.08.06.606880. doi: 10.1101/2024.08.06.606880. bioRxiv. 2024. Update in: Nat Metab. 2025 Jun;7(6):1204-1222. doi: 10.1038/s42255-025-01297-8. PMID: 39149397 Free PMC article. Updated. Preprint.
Comment in
-
Burning fat with cysteine depletion.Cell Stress. 2025 Nov 13;9:216-221. doi: 10.15698/cst2025.11.313. eCollection 2025. Cell Stress. 2025. PMID: 41281417 Free PMC article.
References
MeSH terms
Substances
Grants and funding
- R01 AG073969/AG/NIA NIH HHS/United States
- U54 AG079759/AG/NIA NIH HHS/United States
- P01AG051459/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- P30 DK127984/DK/NIDDK NIH HHS/United States
- P30 AG066508/AG/NIA NIH HHS/United States
- R00DK1208689/U.S. Department of Health & Human Services | NIH | National Institute of Diabetes and Digestive and Kidney Diseases (National Institute of Diabetes & Digestive & Kidney Diseases)
- P01 AG051459/AG/NIA NIH HHS/United States
- P30 DK045735/DK/NIDDK NIH HHS/United States
- AG068863/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- AG073969/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- R01 AG068863/AG/NIA NIH HHS/United States
- AG031797/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- R01 AG031797/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

