This is a preprint.
Aperiodic neural timescales in prefrontal cortex dilate with increased task abstraction
- PMID: 40462968
- PMCID: PMC12132356
- DOI: 10.1101/2025.04.21.649913
Aperiodic neural timescales in prefrontal cortex dilate with increased task abstraction
Abstract
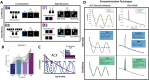
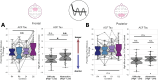
Navigating everyday environments requires that the brain perform information processing at multiple different timescales. For example, while watching a movie we use sensory information from every video frame to construct the current movie scene, which itself is continuously integrated into the narrative arc of the film. This critical function is supported by sensory inputs propagating from dynamic sensory cortices to association cortices, where neural activity remains more stable over time. The hierarchical organization of cortex is therefore reflected in a gradient of neural timescales. While this propagation of inputs up the cortical hierarchy is facilitated by both rhythmic (oscillatory) and non-rhythmic (aperiodic) neural activity, traditional measures of oscillations are often confounded by the influence of aperiodic signals. The reverse is also true: traditional measures of aperiodic neural timescales are influenced by oscillations. This makes it difficult to distinguish between oscillatory and timescale effects in cognition. Here, we analyzed electroencephalography (EEG) data from participants performing a cognitive control task that manipulated the amount of task-relevant contextual information, called task abstraction. Critically, we separated aperiodic neural timescales from the confounding influence of oscillatory power. We hypothesized that neural timescales would increase during the task, and more so in high-abstraction conditions. We found that task abstraction dilated the aperiodic neural timescale, as estimated from the autocorrelation function, over prefrontal cortical regions. Our findings suggests that neural timescales are a dynamic feature of the cerebral cortex that change to meet task demands.
Conflict of interest statement
Competing interests The authors declare no competing interests.
Figures


Similar articles
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Peripuberty Is a Sensitive Period for Prefrontal Parvalbumin Interneuron Activity to Impact Adult Cognitive Flexibility.Dev Neurosci. 2025;47(2):127-138. doi: 10.1159/000539584. Epub 2024 Jun 3. Dev Neurosci. 2025. PMID: 38830346 Free PMC article.
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Antidepressants for pain management in adults with chronic pain: a network meta-analysis.Health Technol Assess. 2024 Oct;28(62):1-155. doi: 10.3310/MKRT2948. Health Technol Assess. 2024. PMID: 39367772 Free PMC article.
References
-
- Shinn M, Hu A, Turner L, Noble S, Preller KH, Ji JL, et al. Functional brain networks reflect spatial and temporal autocorrelation. Nat Neurosci. 2023. May;26(5):867–78. - PubMed
-
- Huntenburg JM, Bazin PL, Margulies DS. Large-Scale Gradients in Human Cortical Organization. Trends Cogn Sci. 2018. Jan 1;22(1):21–31. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
