This is a preprint.
APOE4-induced patterned behavioral decline and neurodegeneration requires endogenous tau in a C. elegans model of Alzheimer's disease
- PMID: 40463229
- PMCID: PMC12132384
- DOI: 10.1101/2025.05.06.652574
APOE4-induced patterned behavioral decline and neurodegeneration requires endogenous tau in a C. elegans model of Alzheimer's disease
Abstract
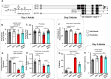
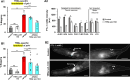
Alzheimer's disease (AD) causes a characteristic spatiotemporal pattern of neurodegeneration, resulting in the loss of associated faculties such as cognition. The factors which account for this pattern of degeneration are unclear, as AD risk genes are numerous and often broadly expressed. Previously, we generated a model of AD using the nematode Caenorhabditis elegans in which the AD risk variant of apolipoprotein E, APOE4, is pan-neuronally expressed. We showed that HSN class motor neurons degenerate in early adult. Here, we expand on our past work by performing behavioral analyses to deduce the effect of APOE4 on the function of distinct neuronal circuits. We found evidence that APOE4 induces dysfunction of other neurons; this spatiotemporal pattern of degeneration roughly correlates with endogenous levels of PTL-1, the C. elegans homolog of human MAPT also known as tau. Moreover, deletion of ptl-1 suppressed defects in multiple behaviors, suggesting broad protective effects across the nervous system including the HSN neurons. Lastly, we show that PTL-1 in the touch receptor neurons, where PTL-1 is most abundant, is required cell nonautonomously for degeneration of the HSN neurons. Our results suggest that C. elegans may provide a useful in vivo system to study how endogenous Tau acts downstream of APOE4 to cause progressive, patterned neurodegeneration.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures


Similar articles
-
APP-Induced Patterned Neurodegeneration Is Exacerbated by APOE4 in Caenorhabditis elegans.G3 (Bethesda). 2020 Aug 5;10(8):2851-2861. doi: 10.1534/g3.120.401486. G3 (Bethesda). 2020. PMID: 32580938 Free PMC article.
-
Differential Signaling Mediated by ApoE2, ApoE3, and ApoE4 in Human Neurons Parallels Alzheimer's Disease Risk.J Neurosci. 2019 Sep 11;39(37):7408-7427. doi: 10.1523/JNEUROSCI.2994-18.2019. Epub 2019 Jul 22. J Neurosci. 2019. PMID: 31331998 Free PMC article.
-
PTL-1 regulates neuronal integrity and lifespan in C. elegans.J Cell Sci. 2013 May 1;126(Pt 9):2079-91. doi: 10.1242/jcs.jcs124404. Epub 2013 Mar 22. J Cell Sci. 2013. PMID: 23525010 Free PMC article.
-
APOE and Alzheimer's Disease: Evidence Mounts that Targeting APOE4 may Combat Alzheimer's Pathogenesis.Mol Neurobiol. 2019 Apr;56(4):2450-2465. doi: 10.1007/s12035-018-1237-z. Epub 2018 Jul 21. Mol Neurobiol. 2019. PMID: 30032423 Review.
-
Use of Caenorhabditis elegans as a model to study Alzheimer's disease and other neurodegenerative diseases.Front Genet. 2014 Sep 5;5:279. doi: 10.3389/fgene.2014.00279. eCollection 2014. Front Genet. 2014. PMID: 25250042 Free PMC article. Review.
References
-
- Andrews-Zwilling Y., Bien-Ly N., Xu Q., Li G., Bernardo A., Yoon S. Y., Zwilling D., Yan T. X., Chen L., and Huang Y. (2010). Apolipoprotein E4 causes age- and Tau-dependent impairment of GABAergic interneurons, leading to learning and memory deficits in mice. The Journal of neuroscience : the official journal of the Society for Neuroscience, 30(41), 13707–13717. 10.1523/JNEUROSCI.4040-10.2010 - DOI - PMC - PubMed
-
- Arnold M. L., Cooper J., Grant B. D., & Driscoll M. (2020). Quantitative Approaches for Scoring in vivo Neuronal Aggregate and Organelle Extrusion in Large Exopher Vesicles in C. elegans. Journal of visualized experiments : JoVE, (163), 10.3791/61368. https://doi.org/10.3791/61368 - DOI - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
