Screening a living biobank identifies cabazitaxel as a strategy to combat acquired taxol resistance in high-grade serous ovarian cancer
- PMID: 40466638
- PMCID: PMC12208324
- DOI: 10.1016/j.xcrm.2025.102160
Screening a living biobank identifies cabazitaxel as a strategy to combat acquired taxol resistance in high-grade serous ovarian cancer
Abstract
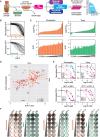
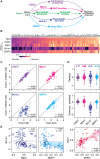
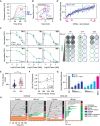
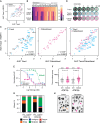
The anti-mitotic agent taxol (paclitaxel) remains a cornerstone of ovarian cancer treatment. To tackle drug resistance and toxicity, second-generation targeted anti-mitotic agents and combination strategies are being explored but have yet to demonstrate meaningful clinical benefits. A limitation is the lack of a platform to compare strategies in models that capture disease heterogeneity. To overcome this, we screen 83 patient-derived ex vivo ovarian cancer models that exhibit extensive intra- and inter-patient heterogeneity, testing four distinct approaches to enhance taxol sensitivity. Inhibitors of the HSET kinesin or the Mps1 spindle assembly checkpoint kinase show minimal impact on the taxol sensitivity landscape. By contrast, Bcl-xL inhibition exerts a global anti-proliferative effect. Inhibition of the MDR1 drug efflux pump restores taxol sensitivity in models characterized by ABCB1 overexpression. These MDR1-driven resistant models also respond to cabazitaxel, which is a poor MDR1 substrate, highlighting a potential therapeutic option for ovarian cancers with acquired taxol resistance.
Keywords: ABCB1; MDR1; centrosome amplification; chromosome instability; drug resistance; intrinsic apoptosis pathway; ovarian cancer; paclitaxel; spindle assembly checkpoint; taxol.
Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures




References
-
- González-Martín A., Harter P., Leary A., Lorusso D., Miller R.E., Pothuri B., Ray-Coquard I., Tan D.S.P., Bellet E., Oaknin A., et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2023;34:833–848. doi: 10.1016/j.annonc.2023.07.011. - DOI - PubMed
-
- Morgan R.D., McNeish I.A., Cook A.D., James E.C., Lord R., Dark G., Glasspool R.M., Krell J., Parkinson C., Poole C.J., et al. Objective responses to first-line neoadjuvant carboplatin-paclitaxel regimens for ovarian, fallopian tube, or primary peritoneal carcinoma (ICON8): post-hoc exploratory analysis of a randomised, phase 3 trial. Lancet Oncol. 2021;22:277–288. doi: 10.1016/s1470-2045(20)30591-x. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

