Daratumumab for PRCA after HCT: study and practical considerations from the EBMT Transplant Complications Working Party
- PMID: 40467580
- PMCID: PMC12137674
- DOI: 10.1038/s41408-025-01315-8
Daratumumab for PRCA after HCT: study and practical considerations from the EBMT Transplant Complications Working Party
Abstract
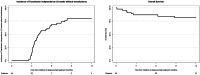
Pure red cell aplasia (PRCA) is a relevant complication after ABO-mismatched allogeneic hematopoietic cell transplantation (HCT). No standard treatment exists, and practice is heterogenous. In this study, we took advantage of an international collaboration to describe characteristics and outcomes of patients receiving daratumumab for PRCA following first allogeneic HCT. We identified 45 patients meeting these criteria (median patient age, 56 years). The median time from HCT to PRCA was 55 days (IQR, 36-116) and all patients were transfusion-dependent at time of daratumumab start. Daratumumab was first-line treatment in 16 patients (36%), most patients (67%) received daratumumab intravenously, and median time from PRCA diagnosis and daratumumab start was 88 days (IQR, 59-219). Incidence of transfusion independence was 69% (95% confidence interval [CI], 52-80%) at 6 months and 80% (95% CI, 62-90%) at 12 months. Incidences of hemoglobin and reticulocyte recoveries were respectively 56 and 78% at 6 months and 65 and 83% at 12 months. Survival at 12 months was 81%, and of 8 deaths, 7 were GVHD- or infection-related. One death was associated with hemolytic anemia. This is the first international and largest study on the use of daratumumab for PRCA after allogeneic HCT, showing high response rates superior to that reported for other treatments. Seven incidents of severe adverse events (mostly infections) underscore the need for close monitoring, proactive management, and comparative studies to determine the role for daratumumab for PRCA. Last, based on these data and a comprehensive literature review, we provide practical consideration for modern PRCA treatment.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures