Production of functional recombinant antibodies in Dictyostelium discoideum
- PMID: 40468350
- PMCID: PMC12139333
- DOI: 10.1186/s13104-025-07314-z
Production of functional recombinant antibodies in Dictyostelium discoideum
Abstract
Objective: Recombinant antibodies are essential reagents for diagnostics, research, and therapy. Numerous production methods have been developed, each of them with its strengths and weaknesses. In this study we evaluated the ability of Dictyostelium discoideum cells to produce and secrete functional antibodies.
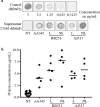
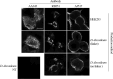
Results: Three recombinant antibodies targeting tubulin, CISD1 or CD8β proteins, respectively, were successfully produced and secreted by D. discoideum cells. Electrophoretic analysis of these antibodies revealed a degradation product, resulting from proteolytic cleavage at the linker peptide connecting the scFv portion to the Fc fragment. Removal of this linker suppressed the proteolytic cleavage. Finally, immunofluorescence analysis confirmed that all three antibodies recognized their target antigen in a specific manner. This study represents the first demonstration that functional recombinant antibodies can be produced in D. discoideum cells.
Keywords: Dictyostelium discoideum; Immunofluorescence; Recombinant antibody production.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not Applicable. Competing interests: The authors declare no competing interests.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

